B. Braun To Showcase at AANA Innovative Portfolio of Products Designed To Help Optimize Regional Anesthesia
06 Setembro 2017 - 2:00PM

B. Braun Medical Inc., a market leader in regional anesthesia, will
present its innovative line of Regional Anesthesia products at the
American Association of Nurse Anesthetists (AANA) conference from
September 8-12, 2017 in Seattle, Washington.
In a simulated hospital setting at booth #428, B. Braun will
showcase the following new products that are bringing a fresh
approach to regional anesthesia:
- Xperius® Point-of-Care Ultrasound System: The
ultrasound system is designed to support the needs in regional
anesthesia at the point of care. The system offers an intuitive
user interface and exceptional image quality for confident needle
targeting and positioning, as well as ergonomic features such as an
articulating arm. Xperius' imaging complements B. Braun's regional
block portfolio. Xperius is the first product resulting from a
strategic alliance between B. Braun Melsungen AG and Royal Philips
(NYSE:PHG) (AEX:PHIA).
- Stimuplex® Ultra 360® Nerve Block
Needles: Stimuplex Ultra 360 needles
feature B. Braun’s new echogenic 360º X-pattern. The ‘X’ is a
unique shape, which when combined with a clear coating, reflects
more ultrasound waves back to the probe than previous designs.1
These needles are designed to provide visibility without
compromising puncture characteristics.
- SAXA Medical Solutions Anesthesia Disinfection
Workstation: B. Braun continues to strive
to reduce healthcare-acquired infections through use of the DOCit™
and HubScrub™ to improve intravascular device disinfection in the
operating room.2 DOCit is designed to disinfect syringe tips and
male luer connectors, and to organize the anesthesia workspace. The
HubScrub is designed to disinfect needleless connectors, female
luers, and swabbable stopcocks. The workstation is designed to
organize the anesthesia workspace in an operating room.
“We are keenly focused on helping clinicians optimize the
regional anesthesia process,” said Peter McGregor, Director of
Marketing, Pain Control, at B. Braun Medical. “We invite attendees
to explore how our products can help improve patient outcomes by
visiting our hospital-like setting at AANA.”
Visitors to B. Braun’s booth at the Washington State Convention
Center also will see the B-Smart™ Injection Pressure Monitor, which
provides a visual indication of high injection pressure and helps
signify intraneural placement of the needle tip during peripheral
nerve blocks.
For more information, visit www.BBraunUSA.com.
About B. Braun B. Braun Medical Inc., a leader
in infusion therapy and pain management, develops, manufactures,
and markets innovative medical products and services to the
healthcare industry. The company is committed to eliminating
preventable treatment errors and enhancing patient, clinician and
environmental safety. B. Braun Medical is headquartered in
Bethlehem, Pa., and is part of the B. Braun Group of Companies in
the U.S., which includes B. Braun Interventional Systems, Aesculap®
and CAPS®.
Globally, the B. Braun Group of Companies employs more than
58,000 employees in 64 countries. Guided by its Sharing Expertise®
philosophy, B. Braun continuously exchanges knowledge with
customers, partners and clinicians to address the critical issues
of improving care and lowering costs. To learn more about B. Braun
Medical, visit www.BBraunUSA.com.
Philips is the manufacturer of the Xperius ultrasound system. B.
Braun Medical Inc. is the distributor of the Xperius ultrasound
system.
*SAXA Medical Solutions, LLC, is the manufacturer of the DOCit™
and HubScrub™ devices. DOCit and HubScrub are registered trademarks
of SAXA Medical Solutions, LLC. B. Braun is a distributor of the
Anesthesia Disinfection Workstation, DOCit and HubScrub.
___________________________________1 Internal testing: ETRs
JMLY-8ADPRZ, NPAK-9VRK2K2 Loftus, R., et al. Reduction in
Intraoperative Bacterial Contamination of Peripheral Intravenous
Tubing Through the Use of a Passive Catheter System,
Anesthesia-Analgesia, December 2012; Volume 115; Number 6.
Contact:
Jason Ford
B. Braun Medical Inc.
610.997.4722
jason.ford@bbraun.com
Koninklijke Philips NV (EU:PHIA)
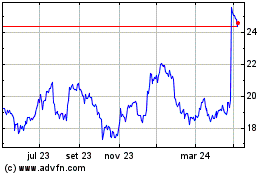
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
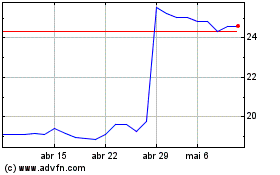
Koninklijke Philips NV (EU:PHIA)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024