Pfizer Completes Acquisition of ReViral
09 Junho 2022 - 11:00AM
Business Wire
- Acquisition expands Pfizer’s anti-infective pipeline and
reinforces commitment to developing both medicines and vaccines to
help combat respiratory syncytial virus (RSV)
Pfizer Inc. (NYSE: PFE) today announced the successful
completion of its acquisition of ReViral, a privately held,
clinical-stage biopharmaceutical company focused on discovering,
developing, and commercializing novel antiviral therapeutics that
target respiratory syncytial virus (RSV).
ReViral brings to Pfizer a portfolio of promising therapeutic
candidates, including sisunatovir, an orally administered inhibitor
designed to block fusion of the RSV virus to the host cell.
Sisunatovir has been granted Fast Track designation by the U.S.
Food and Drug Administration (FDA). It significantly reduced viral
load in a phase 2 RSV human challenge study in healthy adults and
is currently in phase 2 clinical development in infants. The
development program for sisunatovir is expected to continue in both
adult and pediatric populations. A second program is focused on the
inhibition of RSV replication targeting the viral N protein. The
lead candidate in this program is currently in phase 1 clinical
development.
“We are excited to bring ReViral’s promising investigational
treatments for RSV into our anti-infective pipeline at Pfizer. This
acquisition further demonstrates our commitment to advancing
pioneering science – both through our in-house expertise and our
work with leading, innovative companies – with the goal of
delivering new breakthroughs to patients suffering from serious
infectious diseases,” said Mikael Dolsten, M.D., Ph.D., Chief
Scientific Officer and President, Worldwide Research, Development
and Medical of Pfizer. “We believe these therapeutic candidates –
and the scientific expertise that has advanced their development –
will complement our ongoing work to help combat RSV infections, and
we look forward to welcoming our new colleagues to further support
these endeavors.”
RSV is a respiratory pathogen, which can lead to severe and
life-threatening lower respiratory tract infections (LRTIs) in
high-risk populations, including young children, immunocompromised
individuals, and older adults. It is estimated to cause infections
in approximately 64 million people, resulting in about 160,000
deaths, globally each year. Currently, treatment options for RSV
are limited, with care management focused primarily on supportive
measures for people with the illness.
Additional Transaction Details
Under the terms of the agreement, Pfizer acquired ReViral for a
total consideration of up to $525 million, including upfront and
development milestones. If successful, Pfizer believes annual
revenue for these programs has the potential to reach or exceed
$1.5 billion.
For additional background on the acquisition, please read the
announcement press release here.
About Pfizer: Breakthroughs That Change Patients’
Lives
At Pfizer, we apply science and our global resources to bring
therapies to people that extend and significantly improve their
lives. We strive to set the standard for quality, safety and value
in the discovery, development and manufacture of health care
products, including innovative medicines and vaccines. Every day,
Pfizer colleagues work across developed and emerging markets to
advance wellness, prevention, treatments and cures that challenge
the most feared diseases of our time. Consistent with our
responsibility as one of the world's premier innovative
biopharmaceutical companies, we collaborate with health care
providers, governments and local communities to support and expand
access to reliable, affordable health care around the world. For
more than 170 years, we have worked to make a difference for all
who rely on us. We routinely post information that may be important
to investors on our website at www.Pfizer.com. In addition, to
learn more, please visit us on www.Pfizer.com and follow us on
Twitter at @Pfizer and @Pfizer News, LinkedIn, YouTube and like us
on Facebook at Facebook.com/Pfizer.
Disclosure Notice
The information contained in this release is as of June 9, 2022.
Pfizer assumes no obligation to update forward‐looking statements
contained in this release as the result of new information or
future events or developments.
This release contains forward-looking information about Pfizer’s
acquisition of ReViral, ReViral’s pipeline portfolio of therapeutic
candidates, including sisunatovir, and potential revenue, and
Pfizer’s infectious disease research and development and innovative
anti-infective and vaccine portfolio and pipeline, including
Pfizer’s investigational RSV vaccine programs, including their
potential benefits, that involve substantial risks and
uncertainties that could cause actual results to differ materially
from those expressed or implied by such statements. Risks and
uncertainties include, among other things, risks related to the
ability to realize the anticipated benefits of the acquisition,
including the possibility that the expected benefits from the
acquisition will not be realized or will not be realized within the
expected time period; the risk that the businesses will not be
integrated successfully; negative effects of the consummation of
the acquisition on the market price of Pfizer’s common stock and/or
operating results; significant transaction costs; unknown
liabilities; the risk of litigation and/or regulatory actions
related to the acquisition of ReViral’s business; other business
effects and uncertainties, including the effects of industry,
market, business, economic, political or regulatory conditions;
future exchange and interest rates; changes in tax and other laws,
regulations, rates and policies; future business combinations or
disposals; the uncertainties inherent in research and development,
including the ability to meet anticipated clinical endpoints,
commencement and/or completion dates for clinical trials,
regulatory submission dates, regulatory approval dates and/or
launch dates, as well as the possibility of unfavorable new
clinical data and further analyses of existing clinical data; risks
associated with interim data; the risk that clinical trial data are
subject to differing interpretations and assessments by regulatory
authorities; whether regulatory authorities will be satisfied with
the design of and results from the clinical studies; whether and
when drug or biologic license applications may be filed in any
jurisdictions for sisunatovir, Pfizer’s investigational RSV vaccine
or any other investigational products; whether and when any such
applications may be approved by regulatory authorities, which will
depend on myriad factors, including making a determination as to
whether the product's benefits outweigh its known risks and
determination of the product's efficacy and, if approved, whether
sisunatovir, Pfizer’s investigational RSV vaccine or any such other
products will be commercially successful; decisions by regulatory
authorities impacting labeling, manufacturing processes, safety
and/or other matters that could affect the availability or
commercial potential of sisunatovir, Pfizer’s investigational RSV
vaccine or any such other products; uncertainties regarding the
ability to obtain recommendations from vaccine advisory or
technical committees and other public health authorities regarding
Pfizer’s investigational RSV vaccine and uncertainties regarding
the commercial impact of any such recommendations; uncertainties
regarding the impact of COVID-19; and competitive developments.
A further description of risks and uncertainties can be found in
Pfizer’s Annual Report on Form 10-K for the fiscal year ended
December 31, 2021 and in its subsequent reports on Form 10-Q,
including in the sections thereof captioned “Risk Factors” and
“Forward-Looking Information and Factors That May Affect Future
Results”, as well as in its subsequent reports on Form 8-K, all of
which are filed with the U.S. Securities and Exchange Commission
and available at www.sec.gov and www.pfizer.com.
Category: Investments
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220608006177/en/
Media Contact: PfizerMediaRelations@pfizer.com +1 (212) 733-1226
Investor Contact: IR@Pfizer.com +1 (212) 733-4848
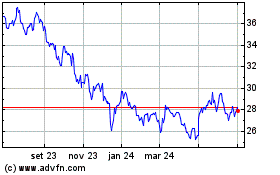
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
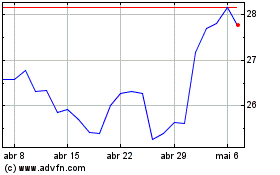
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024