- Dr. Nuyten will oversee Arcus’s clinical development
organization that includes nearly 200 employees and six
clinical-stage programs targeting TIGIT, the adenosine axis (CD73
and dual A2a/A2b), HIF-2a and PD-1
Arcus Biosciences, Inc. (NYSE:RCUS), a clinical-stage, global
biopharmaceutical company focused on developing differentiated
molecules and combination therapies for people with cancer, today
announced that Dimitry S.A. Nuyten, M.D., Ph.D has been appointed
chief medical officer (CMO) effective August 1, 2022. In his role
as CMO, Dr. Nuyten will oversee Arcus’s clinical development
organization that includes nearly 200 employees and six
clinical-stage programs targeting TIGIT, the adenosine axis (CD73
and dual A2a/A2b), HIF-2a and PD-1. Dr. Nuyten will oversee the
advancement of four registrational Phase 3 trials that are ongoing
or expected to start by year end for the anti-TIGIT antibody
domvanalimab in novel combinations across multiple cancers.
“Dr. Nuyten’s attributes and experiences are well matched to
Arcus’s rapid evolution as we enter 2023 with four ongoing
registrational trials for domvanalimab-based combinations and a
portfolio of Phase 2 and early signal-seeking trials investigating
combinations of our six clinical molecules,” said Terry Rosen,
Ph.D., chief executive officer of Arcus. “His proven abilities to
grow and lead large cross-functional teams, interface with
commercial and regulatory organizations and navigate complex
portfolio management will complement our exceptional development
organization and facilitate the continued growth of Arcus. In Dr.
Nuyten’s new role, he will also be working closely with our global
partners, including Gilead Sciences, to optimize and implement our
clinical development strategy.”
As part of his role as CMO, Dr. Nuyten will serve as a member of
Arcus’s executive committee and co-chair of Arcus’s and Gilead’s
Joint Steering Committee. He will be responsible for our clinical
organization, including clinical development and operations,
clinical pharmacology and biometrics. Prior to joining Arcus, Dr.
Nuyten served as senior vice president and CMO of Nektar
Therapeutics where he led a 200-person development organization
which included clinical development, safety, clinical operations,
medical affairs, clinical and non-clinical pharmacology, biostats,
data management and programming. Prior to Nektar, he served as CMO
of Aduro Biotech and served as the immuno-oncology development
leader and vice president of global product development for
oncology at Pfizer, where he led the late-stage development of
Bavencio® and early-stage clinical programs for oncology and
immune-oncology. Prior to Pfizer, Dr. Nuyten was group medical
director at BristolMyers Squibb. He holds a Ph.D. in cancer biology
from the University of Amsterdam Medical School in The Netherlands,
is Board Certified in radiation oncology and certified as a
physician in The Netherlands by the University of Groningen Medical
School. Over the course of his career, Dr. Nuyten has authored
numerous peer-reviewed papers, is co-inventor on multiple patents
and has been recognized with prestigious awards, including as a
two-time recipient of the American Society of Clinical Oncology
Merit Award.
“My career has been dedicated to understanding and exploiting
the biologic drivers of cancer to develop new treatments that have
the potential to improve outcomes or even cure cancer,” said
Dimitry Nuyten, M.D., Ph.D, incoming chief medical officer at Arcus
Biosciences. “I was attracted to Arcus by the breadth and diversity
of its portfolio of molecules and clinical programs and the
corresponding opportunity to substantially impact the way that many
important cancers are treated. I am thrilled to join the company
and excited to work with the talented and patient-centric Arcus
team to translate an innovative pipeline into clinically meaningful
therapies for a broad array of cancers with high unmet medical
need.”
Arcus Ongoing and Announced Clinical Studies
Trial Name
Arms
Setting
Status
NCT No.
Lung Cancer
ARC-7
zim vs. dom + zim vs. etruma +
dom + zim
1L NSCLC (PD-L1 ≥ 50%)
Ongoing Randomized Phase 2
NCT04262856
PACIFIC-8
(AZ)
dom + durva vs. durva
Curative-Intent Stage 3 NSCLC
Ongoing Registrational Phase
3
NCT05211895
ARC-10
dom + zim vs. zim vs. chemo
1L NSCLC (PD-L1 ≥ 50%)
Ongoing Registrational Phase
3
NCT04736173
STAR-121
(GILD)
dom + zim + chemo vs pembro +
chemo
1L NSCLC (PD-L1 all-comers)
Planned Registrational Phase
3
TBD
EDGE-Lung
dom + zim +/- quemli
1L/2L NSCLC (lung cancer platform
study)
In Planning Phase 2
TBD
Lung Platform (GILD)
dom + zim +/- etruma or
sacituzumab govitecan (Trodelvy) or other combos
1L/2L NSCLC (lung cancer platform
study)
In Planning Phase 2
TBD
Gastrointestinal Cancers
ARC-9
etruma + zim + mFOLFOX vs.
SOC
2L/3L/3L+ CRC
Ongoing
Randomized Phase 2
NCT04660812
ARC-21
dom + zim ± chemo
1L/2L Upper GI Malignancies
Ongoing
Phase 2
NCT05329766
STAR-221
dom + zim + chemo vs. nivo +
chemo
GI Malignancies
Planned Registrational Phase
3
TBD
Pancreatic Cancer
ARC-8
quemli + zim + gem/nab-pac vs.
quemli + gem/nab-pac
1L, 2L PDAC
Ongoing Randomized Phase 1/1b
NCT04104672
Prostate Cancer
ARC-6
etruma + zim + SOC vs. SOC
(Adding sacituzumab govitecan (Trodelvy) combination cohorts)
2L/3L CRPC
Ongoing Randomized Phase 2
NCT04381832
Various
ARC-12
AB308 + zim
Advanced Malignancies
Ongoing
Phase 1/1b
NCT04772989
ARC-14
AB521
Healthy Volunteers
Ongoing
NCT05117554
ARC-20
AB521
Cancer Patients / ccRCC
Planned Phase 1/1b
TBD
dom: domvanalimab; durva: durvalumab; etruma: etrumadenant;
gem/nab-pac: gemcitabine/nab-paclitaxel; nivo: nivolumab; pembro:
pembrolizumab; quemli: quemliclustat; SOC: standard of care; zim:
zimberelimab; ccRCC: clear-cell renal cell carcinoma
About the Gilead Collaboration
In May 2020, Gilead and Arcus entered into a 10-year
collaboration that provided Gilead immediate rights to zimberelimab
and the right to opt into all other Arcus programs arising during
the collaboration term. In November 2021, Gilead and Arcus amended
the collaboration in connection with Gilead’s option exercise for
three of Arcus’s then-clinical stage programs. For all other
programs that are in clinical development or new programs that
enter clinical development thereafter, the opt-in payments are $150
million per program. Gilead’s option, on a program-by-program
basis, expires after a specified period of time following the
achievement of a development milestone for such program and Arcus’s
delivery to Gilead of the requisite qualifying data package.
Concurrent with the May 2020 collaboration agreement, Gilead and
Arcus entered into a stock purchase agreement under which Gilead
made a $200 million equity investment in Arcus. That stock purchase
agreement was amended and restated in February 2021 in connection
with Gilead’s increased equity stake in Arcus from 13% to 19.5%,
with an additional $220 million investment.
Gilead and Arcus are co-developing and equally share global
development costs for five clinical candidates, including
domvanalimab, an Fc-silent anti-TIGIT antibody, etrumadenant, a
dual adenosine A2a/A2b receptor antagonist, quemliclustat, a small
molecule inhibitor of CD73, and zimberelimab, an anti-PD1
antibody.
About Arcus Biosciences
Arcus Biosciences is a clinical-stage, global biopharmaceutical
company developing differentiated molecules and combination
medicines for people with cancer. In partnership with industry
partners, patients and physicians around the world, Arcus is
expediting the development of first- or best-in-class medicines
against well-characterized biological targets and pathways and
studying novel, biology-driven combinations that have the potential
to help people with cancer live longer. Founded in 2015, the
company has expedited the development of six investigational
medicines into clinical studies, including new combination
approaches that target TIGIT, PD-1, the adenosine axis (CD73 and
dual A2a/A2b receptor) and most recently, HIF-2a. For more
information about Arcus Biosciences’ clinical and pre-clinical
programs, please visit www.arcusbio.com or follow us on
Twitter.
Forward-Looking Statements
This press release contains forward-looking statements. All
statements regarding events or results to occur in the future
contained herein, including, but not limited to, statements
regarding upcoming trials and initiation timelines and continued
growth of Arcus are forward-looking statements reflecting the
current beliefs and expectations of management made pursuant to the
safe harbor provisions of the Private Securities Litigation Reform
Act of 1995. All forward-looking statements involve known and
unknown risks and uncertainties and other important factors that
may cause Arcus’s actual results, performance or achievements to
differ significantly from those expressed or implied by the
forward-looking statements. Factors that could cause or contribute
to such differences include, but are not limited to: difficulties
or delays in initiating or conducting clinical trials due to
difficulties or delays in the regulatory process, enrolling
subjects or manufacturing or supplying product for such clinical
trials, all of which may be exacerbated by the COVID-19 pandemic;
changes in the competitive landscape for Arcus’s programs; the
unexpected emergence of adverse events or other undesirable side
effects; and the inherent uncertainty associated with
pharmaceutical product development and clinical trials. Risks and
uncertainties facing Arcus are described more fully in its
Quarterly Report on Form 10-Q for the quarter ended June 30, 2022,
filed on August 3, 2022 with the SEC. You are cautioned not to
place undue reliance on the forward-looking statements, which speak
only as of the date of this press release. Arcus disclaims any
obligation or undertaking to update, supplement or revise any
forward-looking statements contained in this press release except
to the extent required by law.
The Arcus name and logo are trademarks of Arcus Biosciences,
Inc. All other trademarks belong to their respective owners.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220808005151/en/
Investor Inquiries: Pia Banerjee Head of Investor
Relations & Strategy (617) 459-2006 pbanerjee@arcusbio.com
Media Inquiries: Holli Kolkey VP of Corporate
Communications (650) 922-1269 hkolkey@arcusbio.com
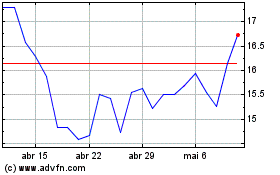
Arcus Biosciences (NYSE:RCUS)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
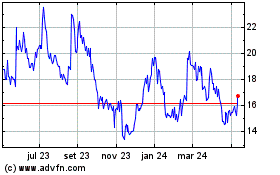
Arcus Biosciences (NYSE:RCUS)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024