France : EIB and MedinCell Sign a New €40 Million Loan Agreement to Support Development of Innovative Treatments
23 Novembro 2022 - 2:00PM
Business Wire
- The 40 million euros loan is intended for the development of
treatments based on MedinCell’s proprietary long-acting injectable
technology aiming at significantly enhance patient care and quality
of life
- This EIB investment is guaranteed by the European Fund for
Strategic Investments, the central pillar of the Investment Plan
for Europe
The new loan signed between the European Investment Bank (EIB),
backed by the Investment Plan for Europe, and pharmaceutical
company MedinCell (Paris:MEDCL) is intended to support the
development of the company’sportfolio of innovative treatments of
the company. MedinCell’s portfolio is already composed of several
Long-Active injectable products at preclinical and clinical stage.
A first product using MedinCell’s BEPO® technology is expected to
reach the U.S. market in the first half of 2023.
Access here the complete press release
About EIB
The EIB is the European Union (EU) long-term financing
institution, and its shareholders are the 27 EU Member States. Its
mission is to contribute to the integration, balanced development,
and economic and social cohesion of EU Member States. It borrows
large volumes of funds from the capital markets and lends them with
very favourable terms to support projects, which contribute to the
achievement of EU objectives. The EIB is working to put the EU at
the forefront of the next wave of innovation, especially in the
health sector. In response to the Covid-19 health crisis, the EIB
has released €6 billion for investments in the health sector to
support medical infrastructure, additional research activities or
other financing related to vaccines and treatments. As a European
bank supporting the climate, the EIB is one of the main fund
providers in the green transition towards a more low-carbon and
sustainable growth model.
About MedinCell
MedinCell is a pharmaceutical company at premarketing stage that
develops a portfolio of long-acting injectable products in various
therapeutic areas by combining its proprietary BEPO® technology
with active ingredients already known and marketed. Through the
controlled and extended release of the active pharmaceutical
ingredient, MedinCell makes medical treatments more efficient,
particularly thanks to improved compliance, i.e. compliance with
medical prescriptions, and to a significant reduction in the
quantity of medication required as part of a one-off or chronic
treatment. The BEPO® technology makes it possible to control and
guarantee the regular delivery of a drug at the optimal therapeutic
dose for several days, weeks or months starting from the
subcutaneous or local injection of a simple deposit of a few
millimeters, fully bioresorbable. MedinCell collaborate with tier
one pharmaceuticals companies and foundations to improve Global
Health through new therapeutic options. Based in Montpellier,
MedinCell currently employs more than 150 people representing over
30 different nationalities. www.medincell.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20221123005399/en/
Press
Medincell: David Heuzé, david.heuze@medincell.com,
+33 6 98 52 47 50
European investment bank : Christophe Alix,
c.alix@eib.org, +33 6 11 81 30 99 www.eib.org/press - press office
: +352 4379 21000 – presse@eib.org
European Commission Flora Matthaes,
flora.matthaes@ec.europa.eu, +32 2 298 39 51
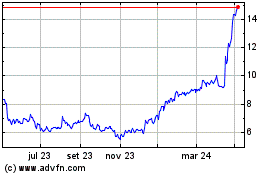
Medincell (EU:MEDCL)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
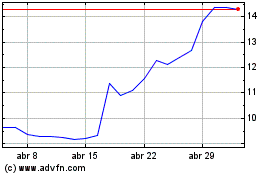
Medincell (EU:MEDCL)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024