Canada Decision on Oral Covid-19 Drugs Likely Weeks Away
07 Janeiro 2022 - 2:39PM
Dow Jones News
By Paul Vieira
OTTAWA-Canada's Health Minister said the country was weeks away
from issuing decisions regarding regulatory authorization for the
use of experimental, oral Covid-19 drugs.
Jean-Yves Duclos made the remarks at a press briefing in Ottawa
on Friday. Merck & Co. and Pfizer Inc. filed late last year
submissions for authorization of the oral drugs. The Federal Drug
Administration last month authorized the use of those companies'
oral Covid-19 antivirals.
The pills are for the newly infected who are considered at high
risk for severe disease, or those who have another medical
condition like cancer, diabetes or a chronic lung disease. Their
use could help cut down infections that require hospital care.
Mr. Duclos acknowledged at the press conference that hospital
capacity in Canada is "stretched too thin," and that's been
exacerbated by a sharp rise in cases due to the Covid-19 Omicron
variant.
On Dec. 3, Canada said it signed deals to acquire 500,000
courses of Merck's Covid-19 oral antiviral treatment, molnupiravir,
with options for an additional 500,000 courses; and an initial
quantity of 1 million courses from Pfizer of its Covid-19 oral
antiviral treatment, called Paxlovid.
Write to Paul Vieira at paul.vieira@wsj.com
-0-
Write to at
(END) Dow Jones Newswires
January 07, 2022 12:24 ET (17:24 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
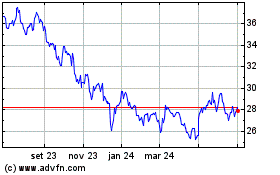
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
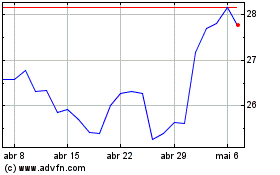
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024