Moderna, Merck Combined Treatment Gets EMA Designation for Melanoma
06 Abril 2023 - 6:53PM
Dow Jones News
By Denny Jacob
Pharmaceutical company Moderna Inc. on Thursday said its
mRNA-4157/V940 in combination with drugmaker Merck & Co.'s
Keytruda was granted priority medicines scheme designation by the
European Medicines Agency to treat patients with high-risk stage
III/IV melanoma following complete resection.
The companies said the designation was based on positive data
from a Phase 2b trial. They plan to initiate a Phase 3 study this
year and expand to additional tumor types, including non-small cell
lung cancer.
Moderna's mRNA-4157/V940 is its investigational personalized
mRNA cancer vaccine. Merck's Keytruda is its anti-PD-1 therapy.
"There is a high unmet need for therapies in melanoma, as it can
be a life-threatening condition where available therapies may not
be sufficiently effective in a significant proportion of patients,"
said Moderna President Stephen Hoge.
Write to Denny Jacob at denny.jacob@wsj.com
(END) Dow Jones Newswires
April 06, 2023 17:38 ET (21:38 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
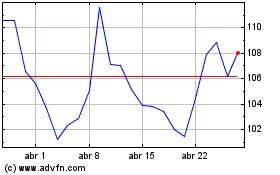
Moderna (NASDAQ:MRNA)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
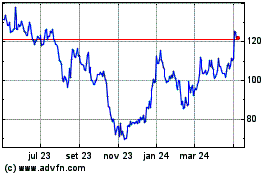
Moderna (NASDAQ:MRNA)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024