MIMEDX Announces Launch of AMNIOEFFECT™
19 Setembro 2022 - 8:00AM

MiMedx Group, Inc. (Nasdaq: MDXG) (“MIMEDX” or the “Company”), a
transformational placental biologics company, today announced the
launch of AMNIOEFFECT™, a tri-layer PURION® processed human tissue
allograft consisting of the amnion, intermediate, and chorion
membrane layers of the placenta.
AMNIOEFFECT complements and expands the
Company’s Surgical Recovery product portfolio. In addition,
AMNIOEFFECT offers superior handling characteristics and provides
surgeons the capability to secure the tissue in place with sutures
when needed to address a surgical wound. During the Limited Market
Release (LMR) phase, AMNIOEFFECT was used in over 50 surgical
cases. Early experience has shown that the product’s handling
characteristics, thickness and broad size availability are
important in large debridement, limb salvage, lumbar fusion and
gynecological procedures.
“The Surgical Recovery market is an engine of
growth for MIMEDX,” said Rohit Kashyap, Ph.D., MIMEDX President,
Wound Care & Surgical. “Our LMR experience with more than 20
surgeons confirmed that AMNIOEFFECT services tangible unmet needs.
I am encouraged by the strong level of physician interest the
product has generated and confident that AMNIOEFFECT will play a
defined role in the physician tool kit. We are committed to
broadening our product offering to drive deeper penetration and
achieve above-market growth.”
“I am optimistic about the potential utility of
AMNIOEFFECT within an array of surgical procedures,” said Dr.
Nicolas Mouawad, Chief of Vascular & Endovascular Surgery,
McLaren Health System – Bay Region, Bay City, Michigan and a
participant in the AMNIOEFFECT LMR. “The field of placental tissue
holds tremendous promise for physicians in need of advanced
treatment options for patients presenting with multiple
comorbidities that can contribute to surgical complications,
delayed healing, and added cost to the healthcare system. Given the
product’s thicker configuration and availability in larger sizes,
we will be able to treat patients needing greater coverage more
effectively and efficiently in the operating room, particularly
those with difficult-to-close surgical wounds.”
About AMNIOEFFECT
AMNIOEFFECT is a tri-layer PURION processed
placental tissue allograft that acts as a semi-permeable barrier,
supporting the healing cascade by aiding the development of
granulation tissue; the tissue provides a biocompatible human
extracellular matrix (ECM) and retains more than 300 regulatory
proteins. AMNIOEFFECT is available in a variety of size
configurations and can be broadly used within procedures requiring
a barrier membrane, such as procedures with: small to large surface
areas; deeper surgical sites; soft tissue defects; tendon, bone, or
hardware exposure; and minimally invasive surgical procedures.
About MIMEDX
MIMEDX is a transformational placental biologics
company, developing and distributing placental tissue allografts
with patent-protected, proprietary processes for multiple sectors
of healthcare. As a pioneer in placental tissue engineering, we
have both a commercial business, focused on addressing the needs of
patients with acute and chronic non-healing wounds, and a promising
late-stage pipeline targeted at decreasing pain and improving
function for patients with degenerative musculoskeletal conditions.
We derive our products from human placental tissues and process
these tissues using our proprietary methods, including the PURION®
process. We employ Current Good Tissue Practices, Current Good
Manufacturing Practices, and terminal sterilization to produce our
allografts. MIMEDX has supplied over two million allografts,
through both direct and consignment shipments. For additional
information, please visit www.mimedx.com.
ContactsInvestors:Jack
HowarthInvestor Relations404.360.5681jhowarth@mimedx.com
Media:Hilary DixonCorporate & Strategic
Communications404.323.4779hdixon@mimedx.com
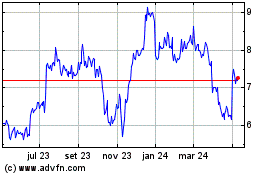
MiMedx (NASDAQ:MDXG)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
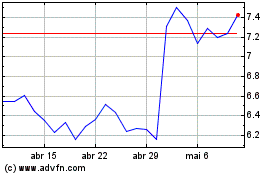
MiMedx (NASDAQ:MDXG)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024