MARLBOROUGH, Mass.,
Dec. 2, 2021 /PRNewswire/ -- Boston
Scientific Corporation (NYSE: BSX) has initiated the MODULAR ATP
clinical trial to evaluate the safety, performance and
effectiveness of the mCRM™ Modular Therapy System. The mCRM System
consists of two cardiac rhythm management (CRM) devices intended to
work together to coordinate therapy: the EMBLEM™ MRI Subcutaneous
Implantable Defibrillator (S-ICD) System and the EMPOWER™ Modular
Pacing System (MPS), which is designed to be the first leadless
pacemaker capable of delivering both bradycardia pacing support and
antitachycardia pacing (ATP).
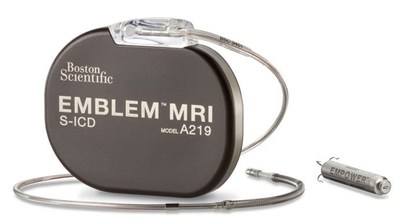
The S-ICD System is a proven treatment option for the prevention
of sudden cardiac death and, because it provides protection without
touching the heart or substernal space, eliminates many of the
complications associated with leads placed in those locations.
Recent data from the PRAETORIAN trial comparing the S-ICD to
transvenous implantable cardioverter defibrillators (TV-ICDs)
confirmed that the S-ICD – which, to date, has been implanted in
more than 100,000 patients worldwide – can be the preferred therapy
choice for the majority of ICD-indicated patients without a need
for pacing.1 Today, patients who may benefit from
receiving an S-ICD System are often implanted with a TV-ICD due to
the possibility of a future need for bradycardia pacing or ATP –
the latter of which is a therapy designed to terminate episodes of
fast, abnormal heart rates.
The EMPOWER MPS, which consists of a leadless pacemaker and
delivery catheter, is designed to complement the S-ICD System by
providing a modular option for patients who develop ATP needs in
the future, potentially expanding the patient population that can
benefit from S-ICD therapy.
"Since the EMPOWER MPS device can be delivered percutaneously
via a minimally invasive approach without the use of leads, the
mCRM System could preserve many of the benefits of the S-ICD System
while offering an option for patients who subsequently develop a
pacing requirement," said Kenneth
Stein, M.D., FACC, FHRS senior vice president and chief
medical officer, Rhythm Management and Global Health Policy, Boston
Scientific. "The components of the system are designed to work in
concert with each other, regardless of when implanted, giving
physicians the ability to provide personalized patient care today
while keeping options open in the future."
The prospective, non-randomized MODULAR ATP clinical trial will
enroll at up to 50 centers in the U.S., Canada and Europe up to 300 patients who are
guideline-indicated for an ICD or who already have an EMBLEM or
EMBLEM MRI S-ICD System implanted. The trial will evaluate the
system- and procedure-related complication-free rate of the EMPOWER
MPS, the communication success between the EMPOWER MPS and the
S-ICD System and adequate pacing capture thresholds of the EMPOWER
MPS.
The first patients in the U.S. were enrolled by Daniel J. Cantillon, M.D., FACC, FHRS, Cleveland
Clinic. Dr. Cantillon, Reinoud Knops, M.D., PhD, Department of
Cardiology, Academic Medical Centre (Amsterdam, Netherlands), Vivek Reddy, M.D., director, Cardiac Arrhythmia
Services, Mount Sinai Hospital; and Prof. Lluis Mont, M.D., PhD, FESC, Arrhythmia section
head, University of Barcelona are
serving as principal investigators of the study.
Preclinical data presented as late-breaking basic science at
American Heart Association Scientific Sessions 2021 demonstrated
that the mCRM System had excellent performance at up to 18 months,
as defined by stable communication between devices, and ability to
deliver therapy as intended.
For more information on the MODULAR ATP trial, visit
www.bostonscientific.com/mcrm. To learn more about the EMBLEM MRI
S-ICD System, visit www.sicdsystem.com.
*The EMPOWER MPS is an investigational device and limited by
U.S. law to investigational use only. It is not available for sale.
Some device references are for informational purposes only and are
pending CE Mark. Not available in the European Economic
Area.
About Boston Scientific
Boston Scientific transforms lives through innovative medical
solutions that improve the health of patients around the world. As
a global medical technology leader for more than 40 years, we
advance science for life by providing a broad range of high
performance solutions that address unmet patient needs and reduce
the cost of healthcare. For more information, visit
www.bostonscientific.com and connect on Twitter and Facebook.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of Section 27A of the Securities Act of 1933 and
Section 21E of the Securities Exchange Act of 1934.
Forward-looking statements may be identified by words like
"anticipate," "expect," "project," "believe," "plan," "estimate,"
"intend" and similar words. These forward-looking statements
are based on our beliefs, assumptions and estimates using
information available to us at the time and are not intended to be
guarantees of future events or performance. These
forward-looking statements include, among other things, statements
regarding our business plans and product performance and
impact. If our underlying assumptions turn out to be
incorrect, or if certain risks or uncertainties materialize, actual
results could vary materially from the expectations and projections
expressed or implied by our forward-looking statements. These
factors, in some cases, have affected and in the future (together
with other factors) could affect our ability to implement our
business strategy and may cause actual results to differ materially
from those contemplated by the statements expressed in this press
release. As a result, readers are cautioned not to place
undue reliance on any of our forward-looking statements.
Factors that may cause such differences include, among other
things: future economic, competitive, reimbursement and regulatory
conditions; new product introductions; demographic trends;
intellectual property; litigation; financial market conditions; and
future business decisions made by us and our competitors. All
of these factors are difficult or impossible to predict accurately
and many of them are beyond our control. For a further list
and description of these and other important risks and
uncertainties that may affect our future operations, see Part I,
Item 1A – Risk Factors in our most recent Annual Report on
Form 10-K filed with the Securities and Exchange Commission, which
we may update in Part II, Item 1A – Risk Factors in
Quarterly Reports on Form 10-Q we have filed or will file
hereafter. We disclaim any intention or obligation to
publicly update or revise any forward-looking statements to reflect
any change in our expectations or in events, conditions or
circumstances on which those expectations may be based, or that may
affect the likelihood that actual results will differ from those
contained in the forward-looking statements. This cautionary
statement is applicable to all forward-looking statements contained
in this document.
CONTACTS:
Laura Aumann
Media Relations
(651) 582-4251 (office)
Laura.Aumann@bsci.com
Lauren Tengler
Investor Relations
(508) 683-4479
BSXInvestorRelations@bsci.com
1 Knops, R. E. et al. Subcutaneous or
transvenous defibrillator therapy. N. Engl. J. Med. 383, 526–536
(2020)
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/boston-scientific-initiates-trial-to-evaluate-industrys-first-modular-crm-system-301436019.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/boston-scientific-initiates-trial-to-evaluate-industrys-first-modular-crm-system-301436019.html
SOURCE Boston Scientific Corporation