Trending: Eisai, Biogen's Alzheimer's Treatment Gets Full FDA Approval
07 Julho 2023 - 1:39AM
Dow Jones News
0408 GMT - Eisai is one of the most mentioned companies in the
news over the past 12 hours, according to Factiva data, after its
Alzheimer's drug Leqembi secured traditional approval from the U.S.
Food and Drug Administration. Leqembi, which the FDA conditionally
approved in January but Medicare didn't widely cover, is part of a
class of Alzheimer's drugs targeting amyloid, a sticky plaque in
the brain some researchers think plays a role in driving the
disease. It is the first anti-amyloid treatment the FDA has fully
approved and the first to clearly slow cognitive decline in
patients with early Alzheimer's. Medicare enrollees will now be
able to get fully covered treatment if they qualify and agree to
report data to a registry. Eisai said Leqembi could generate $7
billion in annual sales globally by 2030. Shares of the Japanese
company were recently 5.6% lower in Tokyo. Dow Jones & Co. owns
Factiva. (jacques.vanwersch@dowjones.com)
(END) Dow Jones Newswires
July 07, 2023 00:24 ET (04:24 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
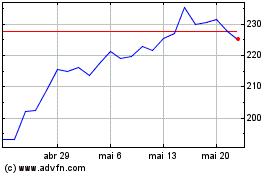
Biogen (NASDAQ:BIIB)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
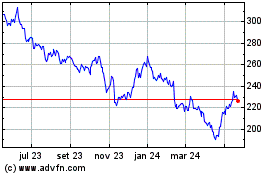
Biogen (NASDAQ:BIIB)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024