Pfizer, BioNTech Get FDA OK for Updated Covid-19 Vaccine
11 Setembro 2023 - 3:23PM
Dow Jones News
By Colin Kellaher
The U.S. Food and Drug Administration has approved an updated
version of the Comirnaty Covid-19 vaccine from Pfizer and BioNTech
that targets a highly contagious sublineage of the virus.
Pfizer and BioNTech on Monday said the FDA approved their
Omicron XBB.1.5-adapted monovalent Covid-19 vaccine for people 12
years and older and granted emergency use authorization for those
ages six months through 11 years.
The vaccine targets an Omicron subvariant called XBB.1.5, which
dominated cases through the spring and summer. A newer subvariant,
called EG.5, currently accounts for the most cases and is closely
related to XBB.1.5, according to the World Health Organization.
Pfizer and BioNTech said preclinical data show their updated
vaccine generates improved neutralizing antibody responses against
multiple Omicron sublineages, including XBB.1.5 and EG.5.1,
unofficially known as Eris.
New York drugmaker Pfizer and Mainz, Germany, immunotherapy
company BioNTech previously said they had been producing the
updated vaccine at risk so it would be ready to ship immediately
upon an FDA green light ahead of an expected rise in demand with
the upcoming fall and winter season.
This companies said the latest vaccine is indicated as a single
dose for most people ages five and older, regardless of prior
Covid-19 vaccination history, while those under the age of five may
be eligible for additional doses if they haven't already completed
a three-dose series with previous formulations.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
September 11, 2023 14:08 ET (18:08 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
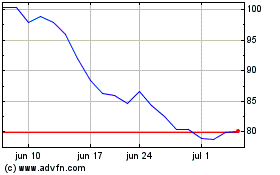
BioNTech (NASDAQ:BNTX)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
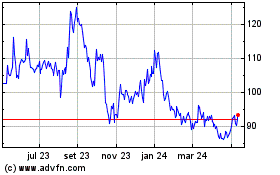
BioNTech (NASDAQ:BNTX)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024