Novavax Stock Drops 13% on Pending FDA Authorization for Updated Covid-19 Vaccine
11 Setembro 2023 - 4:59PM
Dow Jones News
By Sabela Ojea
Shares of Novavax on Monday dropped after the U.S. Food and Drug
Administration authorized for emergency use Moderna and Pfizer's
updated Covid-19 vaccines.
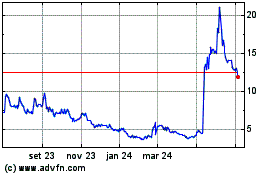
At 15:21 pm E.T., Novavax shares were down 13%, at $7.80. The
stock has dropped 22% since the year started, and 76% in the past
12 months.
The vaccine maker said doses of its updated Covid-19 vaccine
have just arrived in the U.S. and are pending FDA and the Centers
for Disease Control and Prevention action for distribution this
fall.
"We are working closely with the FDA on its review of our
Emergency Use Authorization application," Novavax's Chief Executive
John Jacobs said.
Write to Sabela Ojea at sabela.ojea@wsj.com
(END) Dow Jones Newswires
September 11, 2023 15:44 ET (19:44 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
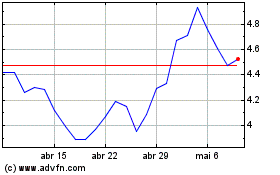
Novavax (NASDAQ:NVAX)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
Novavax (NASDAQ:NVAX)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024