Vanda Pharma Says FDA Accepts New Drug Application for Tradipitant to Treat Gastroparesis
04 Dezembro 2023 - 8:45PM
Dow Jones News
By Stephen Nakrosis
Vanda Pharmaceuticals said the Food and Drug Administration
accepted the filing of the company's new drug application for
tradipitant to treat symptoms of gastroparesis.
Gastroparesis is characterized by delayed gastric emptying with
symptoms including bloating, nausea, vomiting and abdominal pain,
the company said Monday.
Vanda said about 6 million patients in the U.S. have
gastroparesis, many of whom remain undiagnosed.
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
December 04, 2023 18:30 ET (23:30 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
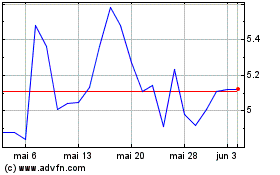
Vanda Pharmaceuticals (NASDAQ:VNDA)
Gráfico Histórico do Ativo
De Mai 2024 até Jun 2024
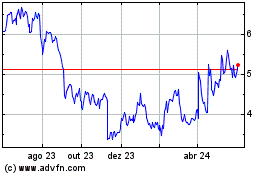
Vanda Pharmaceuticals (NASDAQ:VNDA)
Gráfico Histórico do Ativo
De Jun 2023 até Jun 2024