Chewy Gets FDA Warning About Unapproved Antimicrobial Animal Drugs
14 Dezembro 2023 - 1:34PM
Dow Jones News
By Dean Seal
Chewy and eight other manufacturers and distributors of
antimicrobial animal drugs have received warning letters from the
Food and Drug Administration telling them that those medicines are
unapproved and misbranded.
The agency said on Thursday that it's concerned that using the
products, which contain antimicrobials that are important in human
medicine, without medical oversight contributes to the development
of antimicrobial resistance.
The FDA said it hasn't verified the safety or effectiveness of
the products as part of an animal drug review process or facility
inspections, and that the products are being illegally marketed
over the counter. The products are marketed and labeled for minor
species such as aquarium fish and pet birds, and contain
antimicrobials including amoxicillin and penicillin.
Representatives for Chewy didn't immediately respond to a
request for comment.
Among the other companies that received FDA warning letters were
Kraft Drug and California Veterinary Supply.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
December 14, 2023 11:19 ET (16:19 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
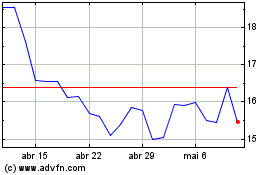
Chewy (NYSE:CHWY)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
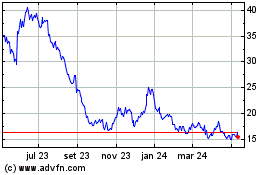
Chewy (NYSE:CHWY)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024