Immutep Doses First Patient in AIPAC-003 Phase II/III Trial for Metastatic Breast Cancer
25 Maio 2023 - 9:00AM

Immutep Limited (ASX: IMM; NASDAQ: IMMP) ("Immutep” or “the
Company”), a clinical-stage biotechnology company developing novel
LAG-3 immunotherapies for cancer and autoimmune disease, today
announces the first patient has been enrolled and safely dosed at a
European clinical site for its integrated Phase II/III AIPAC-003
trial in metastatic breast cancer.
AIPAC-003 is evaluating eftilagimod alpha
(“efti”), Immutep’s soluble LAG-3 protein and first-in-class MHC
Class II agonist, in combination with standard-of-care paclitaxel
for the treatment of metastatic HER2-neg/low breast cancer and
triple-negative breast cancer. It will take place at
approximately 17 clinical sites across Europe and the United States
of America. Patients will receive same-day administration of efti +
paclitaxel that can continue until disease progression.
Immutep
CSO, Prof Frédéric
Triebel said: “Commencing patient dosing for our AIPAC-003
trial of efti is a significant milestone for Immutep. Our aim is to
improve clinical outcomes, focusing on a robust primary endpoint
later in the phase III, overall survival, for patients with
standard-of-care chemotherapy. Our previous trial, AIPAC, showed
encouraging efficacy and safety results, including a 2.9-month
median overall survival benefit and statistically significant
median overall survival improvements of between 4.2 to 19.6 months
across three pre-specified subgroups. We look forward to seeing how
90mg efti dosing, along with same-day administration of efti plus
paclitaxel until disease progression, may build upon these prior
results.”
AIPAC-003 includes an open-label lead-in of up
to 12 patients dosed at 90mg efti, which will be followed by a
randomized (1:1) portion of the Phase II consisting of up to 58
evaluable patients who will receive 30mg efti or 90mg efti to
determine the optimal biological dose in combination with
paclitaxel. Depending on the Phase II results, potential regulatory
actions and resources, a randomized, double-blinded,
placebo-controlled Phase III portion will then follow. The Phase
III will have overall survival as the primary objective and may
include a specific patient population.
About Eftilagimod Alpha (Efti)
Efti is Immutep’s proprietary soluble LAG-3
protein and MHC Class II agonist that stimulates both innate and
adaptive immunity for the treatment of cancer. As a first-in-class
antigen presenting cell (APC) activator, efti binds to MHC (major
histocompatibility complex) Class II molecules on APC leading to
activation and proliferation of CD8+ cytotoxic T cells, CD4+ helper
T cells, dendritic cells, NK cells, and monocytes. It also
upregulates the expression of key biological molecules like IFN-ƴ
and CXCL10 that further boost the immune system’s ability to fight
cancer.
Efti is under evaluation for a variety of solid
tumours including non-small cell lung cancer (NSCLC), head and neck
squamous cell carcinoma (HNSCC), and metastatic breast cancer. Its
favourable safety profile enables various combinations, including
with anti-PD-[L]1 immunotherapy and/or chemotherapy. Efti has
received Fast Track Designation in 1st line HNSCC and in 1st line
NSCLC from the United States Food and Drug Administration
(FDA).
About ImmutepImmutep is a
clinical stage biotechnology company developing novel LAG-3
immunotherapy for cancer and autoimmune disease. We are pioneers in
the understanding and advancement of therapeutics related to
Lymphocyte Activation Gene-3 (LAG-3), and our diversified product
portfolio harnesses its unique ability to stimulate or suppress the
immune response. Immutep is dedicated to leveraging its expertise
to bring innovative treatment options to patients in need and to
maximise value for shareholders. For more information, please visit
www.immutep.com.
Australian
Investors/Media:Catherine Strong, Citadel-MAGNUS+61 (0)406
759 268; cstrong@citadelmagnus.com
U.S. Media:Chris Basta, VP,
Investor Relations and Corporate Communications+1 (631) 318 4000;
chris.basta@immutep.com
Immutep (NASDAQ:IMMP)
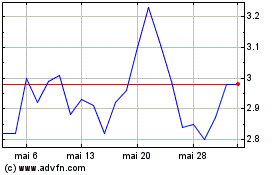
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
Immutep (NASDAQ:IMMP)
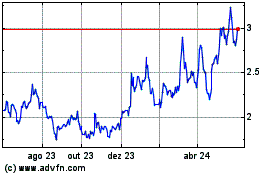
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024