ABVC Signs a Term Sheet to Earn Licensing Fees of up to $20M and Royalties of 5-12% of Net Sales
01 Agosto 2023 - 9:30AM

via NewMediaWire -- ABVC Biopharma, Inc. (NASDAQ: ABVC), a
clinical-stage biopharmaceutical company developing therapeutic
solutions in oncology/hematology, neurology, and ophthalmology,
today announced the signing of a legally binding term sheet with a
prominent Chinese pharmaceutical company, Xinnovation Therapeutics
Co., Ltd that, subject to definitive agreements, is for the
exclusive licensing of ABV-1504 for Major Depressive Disorder (MDD)
and ABV-1505 for Attention-Deficit/Hyperactivity Disorder in
mainland China.
Under this landmark agreement, Xinnovation Therapeutics Co., Ltd
will hold exclusive rights to develop, manufacture, market, and
distribute our innovative drugs for MDD and ADHD in the Chinese
market and shall bear the costs for clinical trials and product
registration in China. The licensing deal carries a possible
aggregate income of $20 million for ABVC. The term sheet outlines
the key terms and conditions of the licensing agreement. The final
agreement, expected to be formally signed within one year, will
specify the precise licensing terms, including the schedule for
milestone payments and other financial arrangements. As per the
term sheet, ABVC expects to receive royalty payments ranging from
5% to 12% based on the projected annual net sales of licensed drugs
in China, which we believe would bring ABVC's revenue ~$50 million
annually and ~$1 billion during the patent life of these products,
if we achieve the expected amount of sales. The final agreement is
subject to the satisfaction of closing conditions, and the
transaction will not occur if they are not satisfied or waived
within one year.
"We are delighted to embark on this partnership with Xinnovation
Therapeutics Co., Ltd., which demonstrates our commitment to
extending the reach of our groundbreaking therapeutics to patients
in China," said Dr. Uttam, CEO of ABVC. "By leveraging Xinnovation
Therapeutics Co., Ltd's strong presence and expertise in the
Chinese market, we aim to provide new hope and improved treatment
options for individuals suffering from MDD and ADHD in China."
Since our neurology products, ABV-1504 and ABV-1505, are new
botanical drugs of the extract from Polygala tenuifolia (Yuanzhi),
a traditional Chinese medicine, Xinnovation will cooperate with
other major pharmaceutical companies in China to grow raw materials
in compliance with GAP (Good Agricultural Practices) and to
manufacture the drug substance in compliance with GMP (Good
Manufacturing Practices). By utilizing the vast agricultural
resources and expertise available in China, we can expedite the
refinement and advancement of these new drugs, paving the way for
faster and more efficient global dissemination.
We believe the Company's pipeline products have great market
potential. As per the Future Market Insights report, the MDD market
was valued at $11.51 billion in 2022 and is expected to reach
$14.96 billion by 2032 with a CAGR of 2.8% over the forecast
period.1 According to the Polaris market research report, the
global ADHD treatment market was valued at $16.13 billion in 2022
and is expected to reach $32.14 billion by 2030 with a CAGR of 7.1%
over the forecast period.2
About ABVC BioPharma
ABVC BioPharma is a clinical-stage biopharmaceutical company
with an active pipeline of six drugs and one medical device
(ABV-1701/Vitargus®) under development. For its drug products, the
Company utilizes in-licensed technology from its network of
world-renowned research institutions to conduct proof-of-concept
trials through Phase II of clinical development. The Company's
network of research institutions includes Stanford University,
University of California at San Francisco, and Cedars-Sinai Medical
Center. For Vitargus®, the Company intends to conduct global
clinical trials through Phase III.
Forward-Looking Statements
This press release contains "forward-looking statements." Such
statements may be preceded by the words "intends," "may," "will,"
"plans," "expects," "anticipates," "projects," "predicts,"
"estimates," "aims," "believes," "hopes," "potential," or similar
words. Forward-looking statements are not guarantees of future
performance, are based on certain assumptions, and are subject to
various known and unknown risks and uncertainties, many of which
are beyond the Company's control, and cannot be predicted or
quantified, and, consequently, actual results may differ materially
from those expressed or implied by such forward-looking statements.
None of the outcomes expressed herein are guaranteed. Such risks
and uncertainties include, without limitation, risks and
uncertainties associated with (i) our inability to manufacture our
product candidates on a commercial scale on our own, or in
collaboration with third parties; (ii) difficulties in obtaining
financing on commercially reasonable terms; (iii) changes in the
size and nature of our competition; (iv) loss of one or more key
executives or scientists; and (v) difficulties in securing
regulatory approval to proceed to the next level of the clinical
trials or to market our product candidates. More detailed
information about the Company and the risk factors that may affect
the realization of forward-looking statements is set forth in the
Company's filings with the Securities and Exchange Commission
(SEC), including the Company's Annual Report on Form 10-K and its
Quarterly Reports on Form 10-Q. Investors are urged to read these
documents free of charge on the SEC's website at
http://www.sec.gov. The Company assumes no obligation to publicly
update or revise its forward-looking statements as a result of new
information, future events or otherwise.
1. https://www.futuremarketinsights.com/reports/major-depressive-disorder-treatment-market#:~:text=The%20major%20depressive%20disorder%20(MDD,US%24%2011.51%20billion%20in%202022. https://www.prnewswire.com/news-releases/global-attention-deficit-hyperactivity-disorder-adhd-market-size-projected-to-reach-usd-32-14-billion-by-2032--with-cagr-of-7-1-study-by-polaris-market-research-301729196.html#:~:text=According%20to%20the%20research%20report,Deficit%20Hyperactivity%20Disorder%20(ADHD)%3F
Contact:Tom MastersonEmail: tmasterson@allelecomms.com
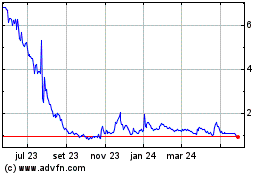
ABVC BioPharma (NASDAQ:ABVC)
Gráfico Histórico do Ativo
De Mai 2024 até Jun 2024
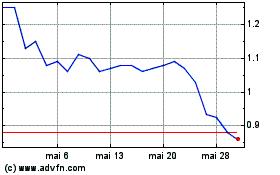
ABVC BioPharma (NASDAQ:ABVC)
Gráfico Histórico do Ativo
De Jun 2023 até Jun 2024