AIM ImmunoTech Reports Second Quarter Financial Results and Provides Clinical Pipeline Update
15 Agosto 2023 - 7:45AM

AIM ImmunoTech Inc. (NYSE American:
AIM) (“AIM” or the “Company”) today reported its
financial results for the second quarter 2023 and provided a
business update. As previously announced, the Company will host a
conference call and audio webcast on Tuesday, August 15, 2023, at
8:30 AM ET (details below).
“Over the course of the second quarter, our team
continued to deliver on our timelines and made significant progress
advancing Ampligen® through our active human clinical studies in
multiple indications. We continue to be encouraged by the growing
body of positive and consistent data and believe Ampligen has the
potential to be a meaningful treatment option across a number of
high-value indications,” commented Thomas K. Equels, Chief
Executive Officer of AIM. “Our operational execution remains
supported by a strong cash position, and we are steadfast in our
mission to advance the clinical development of our oncology
pipeline, with pancreatic cancer as our lead development program.
Looking ahead to the remainder of the year, we believe we are
well-positioned to achieve a number of value-driving
milestones.”
Recent Highlights
- Ampligen identified as one of two
potential therapeutics possessing modest to high potential for the
treatment of post-COVID conditions out of 22 identified on-going
randomized clinical trials in recent peer-reviewed
publication.
- Received updated data from Early
Access Program (EAP) at Erasmus Medical Center which bolsters
previously published data indicating that treatment with Ampligen
following FOLFIRINOX was associated with improved survival rates in
pancreatic cancer patients compared to matched controls of patients
who did not receive Ampligen.
- Commenced and completed full
enrollment in the Company’s Phase 2 study evaluating Ampligen as a
potential therapeutic for people with post-COVID conditions
(AMP-518).
- Announced the publication of
pre-clinical data that suggests Ampligen has the potential to act
directly on tumor cells to reduce tumor cell growth in pancreatic
cancer patients with sufficient tumor levels of TLR-3, suggesting a
potential biomarker to identify patients who may respond to
Ampligen. The anti-tumor analysis was published in the
peer-reviewed journal American Journal of Cancer Research in the
paper “Rintatolimod: A potential treatment in patients with
pancreatic cancer expressing Toll-like receptor 3.”
- Received the required approvals
from the Netherlands for Erasmus Medical Center (“Erasmus MC”) to
begin a Phase 1b/2 study under the previously announced external
sponsored collaborative clinical research agreement with
AstraZeneca and Erasmus MC.
- Announced the opening of an
additional clinical trial site at University of Nebraska for Phase
2 study of Ampligen for the treatment of pancreatic cancer
(AMP-270).
- Granted patent No. 2022/01079,
titled “Compositions and Methods Useful for Ebola Virus Infection”
by the South African Patent and Trademark Office (CIPC).
Ampligen Expected Upcoming Pipeline
Milestones
Q3 2023
- Locally Advanced Pancreatic Cancer:
Enroll and dose first patient in Phase 2 study
- Advanced Recurrent Ovarian Cancer:
Announce protocol-planned interim results
Q4 2023
- Metastatic Pancreatic Cancer:
Enroll and dose first patient in Phase 1b/2 study evaluating
Ampligen in combination with AstraZeneca’s Imfinzi
(durvalumab)
Summary of Financial Highlights for
Second Quarter 2023
- As of June 30, 2023, AIM reported
cash, cash equivalents and marketable securities of $28.4 million.
Based on management’s current expectation, the Company’s cash
runway is expected to fund operations through multiple key
milestones through the end of 2024.
- Research and development expenses
for the three months ended June 30, 2023 were $3.0 million,
compared to $2.5 million for the same period in 2022.
- General and administrative expenses
were $2.6 million for the three months ended June 30, 2023,
compared to $2.2 million for the same period 2022.
- The net loss from operations for
the three months ended June 30, 2023 was $4.9 million, or $0.10 per
share, compared to $4.9 million, or $0.10 per share, for the three
months ended June 30, 2022.
Please refer to the full 10-Q for complete
details.
Conference Call and Webcast Details
As previously announced, the Company will host a
quarterly conference call and live audio webcast to discuss the
operational and financial results on August 15, 2023, at 8:30 AM
ET.
The call will be hosted by members of AIM, CEO
Thomas K. Equels and Scientific Officer Christopher McAleer, PhD.
Interested participants and investors may access the conference
call by dialing (877) 407-9219 (domestic) or (201) 689-8852
(international) and referencing the AIM ImmunoTech Conference Call.
The live webcast will be accessible on the Events page of the
Investors section of the Company’s website, aimimmuno.com, and will
be archived for 90 days following the live event.
About AIM ImmunoTech Inc.
AIM ImmunoTech Inc. is an immuno-pharma company
focused on the research and development of therapeutics to treat
multiple types of cancers, immune disorders and viral diseases,
including COVID-19. The Company’s lead product is a first-in-class
investigational drug called Ampligen® (rintatolimod), a dsRNA and
highly selective TLR3 agonist immuno-modulator with broad spectrum
activity in clinical trials for globally important cancers, viral
diseases and disorders of the immune system.
For more information, please
visit aimimmuno.com and connect with the Company
on Twitter, LinkedIn, and Facebook.
Cautionary Statement
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995 (the “PSLRA”). Words such as “may,” “will,”
“expect,” “plan,” “anticipate” and similar expressions (as well as
other words or expressions referencing future events or
circumstances) are intended to identify forward-looking statements.
Many of these forward-looking statements involve a number of risks
and uncertainties. Among other things, for those statements, the
Company claims the protection of safe harbor for forward-looking
statements contained in the PSLRA. The Company does not undertake
to update any of these forward-looking statements to reflect events
or circumstances that occur after the date hereof.
Investor Contact:
JTC Team, LLC
Jenene Thomas
(833) 475-8247
AIM@jtcir.com
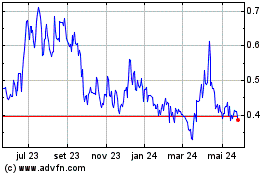
AIM ImmunoTech (AMEX:AIM)
Gráfico Histórico do Ativo
De Ago 2024 até Set 2024
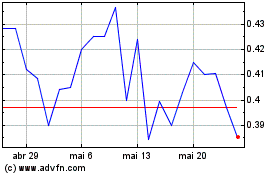
AIM ImmunoTech (AMEX:AIM)
Gráfico Histórico do Ativo
De Set 2023 até Set 2024