AEterna Zentaris Receives Positive Opinion for Orphan Medicinal Product Designation for Perifosine for the Treatment of Multiple
01 Março 2010 - 9:30AM
PR Newswire (US)
QUEBEC CITY, March 1 /PRNewswire-FirstCall/ -- AEterna Zentaris
Inc. (the "Company"), a late-stage drug development company
specialized in oncology and endocrinology, today announced that it
has received a positive opinion for orphan medicinal product
designation for its compound perifosine from the Committee for
Orphan Medicinal Products (COMP) of the European Medicines Agency,
for the treatment of multiple myeloma. Keryx Biopharmaceuticals,
Inc. , AEterna Zentaris' partner and licensee for perifosine in the
North American market, already announced in September 2009 that it
has received Orphan Drug Designation for perifosine for the
treatment of multiple myeloma from the U.S. Food and Drug
Administration (FDA). Juergen Engel, Ph. D., President and CEO of
AEterna Zentaris stated, "We are very pleased with the positive
opinion which is summarized by the COMP as follows: 'The
preclinical data and the preliminary clinical data obtained with
perifosine justifies the claim on a clinically relevant advantage
based in particular on the response obtained in relapsed and
refractory multiple myeloma patients. Furthermore, perifosine is
intended for oral administration, compared to the currently
available parenteral treatments. This supports the assumption for a
major contribution to patient care with regards to convenience and
avoidance of a more aggressive administration route.'" About Orphan
Medicinal Product Designation Orphan medicinal product designation
is granted by the European Commission, following a positive opinion
from the COMP, to a medicinal product that is intended for the
diagnosis, prevention or treatment of a life-threatening or a
chronically debilitating condition affecting not more than five in
10,000 persons in the Community when the application for
designation is submitted. Orphan medicinal product designation
provides the sponsor with access to the Centralized Procedure for
the application for marketing authorization, protocol assistance,
up to a 100% reduction in fees related to a marketing authorization
application, pre-authorization inspection and post-authorization
activities, and could provide ten years of market exclusivity in
EU, once approved for the treatment of multiple myeloma. About
Multiple Myeloma Multiple myeloma, a cancer of the plasma cell, is
an incurable but treatable disease. Multiple myeloma is the second
most-common hematologic cancer, representing 1% of all cancer
diagnoses and 2% of all cancer deaths. According to the
International Myeloma Foundation, more than 85,000 men and women in
Europe were currently undergoing treatment for multiple myeloma in
2007 and 25,000 people were expecting to die from multiple myeloma
in 2007. According to the American Cancer Society, an estimated
20,580 new cases of multiple myeloma were diagnosed in the United
States and 10,500 people were expecting to die from multiple
myeloma in the United States in 2009. To date, several authorized
therapies exist for the treatment of multiple myeloma. Despite this
progress, patients continue to relapse, become refractory to prior
treatments and eventually die from their disease. Thus, new
therapies are needed to treat these patients and extend their
survival. About Perifosine Perifosine is a novel oral anti-cancer
agent that modulates several key signal transduction pathways,
including Akt, MAPK, and JNK that have been shown to be critical
for the survival of cancer cells. Perifosine has demonstrated both
safety and clinical efficacy in several tumor types, both as a
single agent and in combination with novel therapies. A Phase 3
study in multiple myeloma, under Special Protocol Assessment (SPA)
and Fast-track review, was initiated in December 2009 in the U.S.
and recruitment for a Phase 3 trial, also under SPA, in refractory
metastatic colorectal cancer should be started in Q2-2010 in the
U.S. Perifosine is currently also in Phase 2 clinical development
for multiple tumor types. About AEterna Zentaris Inc. AEterna
Zentaris Inc. is a late-stage drug development company specialized
in oncology and endocrinology. News releases and additional
information are available at http://www.aezsinc.com/
Forward-Looking Statements This press release contains
forward-looking statements made pursuant to the safe harbor
provisions of the U.S. Securities Litigation Reform Act of 1995.
Forward-looking statements involve known and unknown risks and
uncertainties, which could cause the Company's actual results to
differ materially from those in the forward-looking statements.
Such risks and uncertainties include, among others, the
availability of funds and resources to pursue R&D projects, the
successful and timely completion of clinical studies, the ability
of the Company to take advantage of business opportunities in the
pharmaceutical industry, uncertainties related to the regulatory
process and general changes in economic conditions. Investors
should consult the Company's quarterly and annual filings with the
Canadian and U.S. securities commissions for additional information
on risks and uncertainties relating to the forward-looking
statements. Investors are cautioned not to rely on these
forward-looking statements. The Company does not undertake to
update these forward-looking statements. We disclaim any obligation
to update any such factors or to publicly announce the result of
any revisions to any of the forward-looking statements contained
herein to reflect future results, events or developments except if
we are required by a governmental authority or applicable law.
DATASOURCE: AETERNA ZENTARIS INC. CONTACT: Investor Relations:
Ginette Vallieres, Investor RelationsCoordinator, (418) 652-8525
ext. 265, gvallieres@aezsinc.com; Media Relations:Paul Burroughs,
Director of Communications, (418) 652-8525 ext.
406,pburroughs@aezsinc.com
Copyright
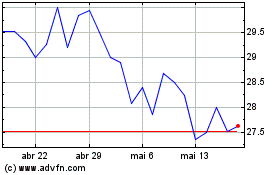
Adams Resources and Energy (AMEX:AE)
Gráfico Histórico do Ativo
De Jun 2024 até Jul 2024
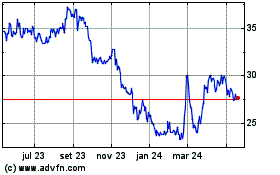
Adams Resources and Energy (AMEX:AE)
Gráfico Histórico do Ativo
De Jul 2023 até Jul 2024