Arcturus Therapeutics Joint Venture mRNA Manufacturing Partner, ARCALIS, Awarded $115 Million in Grants from the Japanese Government
14 Agosto 2023 - 10:00AM
Business Wire
LUNAR-FLU collaboration with CSL Seqirus
achieves additional $17.5 million development milestone
Arcturus Therapeutics Holdings Inc. (the “Company”, “Arcturus”,
Nasdaq: ARCT), a global late-stage clinical messenger RNA medicines
company focused on the development of infectious disease vaccines
and opportunities within liver and respiratory rare diseases, today
announced that ARCALIS Co., Ltd., its manufacturing joint venture
in Japan to support the production of mRNA vaccines and
therapeutics, has been awarded $115 million in two separate grants
from the Japanese government. The grants will be used to fund the
construction of a factory and the purchase of capital equipment to
support current Good Manufacturing Practice (cGMP) production of
mRNA drug substance and mRNA drug product operations.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20230814249381/en/
The ARCALIS building in Japan. (Photo:
Business Wire)
ARCALIS is a joint venture between Axcelead and Arcturus,
wherein Arcturus holds a 49% equity position that is subject to
dilution. The factory, located in Minamisoma City, Japan, is
designed to provide cGMP integrated vaccine manufacturing, from
mRNA drug substance to bulk nanoparticle-formulated drug
product.
“It was an honor to welcome senior Japanese government officials
and dignitaries to the opening ceremonies for ARCALIS’
state-of-the-art factory which will support the cGMP manufacturing
of Arcturus’ next-generation STARR® mRNA vaccines,” said Pad
Chivukula, Ph.D., Chief Scientific Officer of Arcturus
Therapeutics.
Arcturus also announced today that the LUNAR-FLU program
continues to progress with funding and operational support from
CSL-Seqirus, including an additional $17.5 million milestone which
will be used to fund flu program development activities. The
LUNAR-FLU program utilizes Arcturus’ clinically validated
next-generation STARR® mRNA platform technology.
“We are pleased with the progress of the LUNAR-FLU program with
CSL Seqirus, our exclusive global licensee for the development of
novel mRNA vaccines, and the significant role that Arcturus is
playing in the development activities,” said Joseph Payne,
President and CEO of Arcturus Therapeutics. “The LUNAR-FLU program
could represent a major advance in vaccination efforts against flu,
potentially providing higher levels of efficacy and greater
durability compared to current flu vaccine approaches. We look
forward to advancing this promising program.”
About Arcturus Therapeutics
Founded in 2013 and based in San Diego, California, Arcturus
Therapeutics Holdings Inc. (Nasdaq: ARCT) is a global late-stage
clinical mRNA medicines and vaccines company with enabling
technologies: (i) LUNAR® lipid-mediated delivery, (ii) STARR® mRNA
Technology (samRNA) and (iii) mRNA drug substance along with drug
product manufacturing expertise. The Company has ongoing
collaborations with CSL Seqirus and Meiji Seika Pharma, and a joint
venture with ARCALIS. Arcturus’ pipeline includes RNA therapeutic
candidates to potentially treat ornithine transcarbamylase
deficiency and cystic fibrosis, along with its partnered mRNA
vaccine programs for SARS-CoV-2 (COVID-19) and influenza. Arcturus’
versatile RNA therapeutics platforms can be applied toward multiple
types of nucleic acid medicines including messenger RNA, small
interfering RNA, circular RNA, antisense RNA, self-amplifying RNA,
DNA, and gene editing therapeutics. Arcturus’ technologies are
covered by its extensive patent portfolio (patents and patent
applications issued in the U.S., Europe, Japan, China, and other
countries). For more information, visit www.ArcturusRx.com. In
addition, please connect with us on Twitter and LinkedIn.
Forward Looking Statements
This press release contains forward-looking statements that
involve substantial risks and uncertainties for purposes of the
safe harbor provided by the Private Securities Litigation Reform
Act of 1995. Any statements, other than statements of historical
fact included in this press release, are forward-looking
statements, including those regarding strategy, future operations,
the issuance of funds by the Japanese government to ARCALIS, the
anticipated use of the funds by ARCALIS, the potential manufacture
by ARCALIS of Arcturus vaccines, the continued progress of the
LUNAR-FLU program, and the impact of general business and economic
conditions. Arcturus may not actually achieve the plans, carry out
the intentions or meet the expectations or projections disclosed in
any forward-looking statements such as the foregoing and you should
not place undue reliance on such forward-looking statements. These
statements are only current predictions or expectations, and are
subject to known and unknown risks, uncertainties, and other
factors that may cause our or our industry’s actual results, levels
of activity, performance or achievements to be materially different
from those anticipated by the forward-looking statements, including
those discussed under the heading "Risk Factors" in Arcturus’ most
recent Annual Report on Form 10-K, and in subsequent filings with,
or submissions to, the SEC, which are available on the SEC’s
website at www.sec.gov. Except as otherwise required by law,
Arcturus disclaims any intention or obligation to update or revise
any forward-looking statements, which speak only as of the date
they were made, whether as a result of new information, future
events or circumstances or otherwise.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230814249381/en/
IR and Media Contacts Arcturus Therapeutics Neda
Safarzadeh VP, Head of IR/PR/Marketing (858) 900-2682
IR@ArcturusRx.com
Kendall Investor Relations Carlo Tanzi, Ph.D. (617) 914-0008
ctanzi@kendallir.com
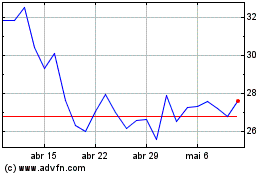
Arcturus Therapeutics (NASDAQ:ARCT)
Gráfico Histórico do Ativo
De Mai 2024 até Jun 2024
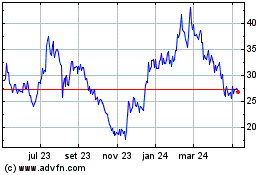
Arcturus Therapeutics (NASDAQ:ARCT)
Gráfico Histórico do Ativo
De Jun 2023 até Jun 2024