electroCore Announces New Reseller Agreement with Red One Medical Devices, LLC.
24 Agosto 2021 - 9:00AM

electroCore, Inc. (Nasdaq: ECOR), a commercial-stage bioelectronic
medicine company, today announced a reseller agreement with Red One
Medical Devices, LLC. (Red One). The partnership will further
expand availability of electroCore’s gammaCore platform in the
federal marketplace.
Red One is a Service-Disabled Veteran-Owned Small Business
(SDVOSB) offering medical and pharmaceutical goods and services to
the U.S. Department of Veterans Affairs (VA) and Department of
Defense (DoD) hospitals, ensuring compliance with contracting
regulations, and providing logistical solutions connected directly
into government contracting portals such as ECAT, GSA, DAPA, BPA,
and DIBBS.“As a VA and DoD private sector partner for medical
innovation, we are excited to partner with electroCore,” said
Charles Pollak, President of Red One. “Their gammaCore therapy may
help thousands of military personnel, family and veterans suffering
from primary headache. Painful and disabling headaches torment more
than 20% of former service members, especially those from the
post-9/11 combat veteran cohort. We look forward to partnering with
electroCore to make the therapy more easily accessible in the
federal marketplace.”“Partnering with Red One will help accelerate
adoption of electroCore’s therapy within the government channel,”
commented Dan Goldberger, Chief Executive Officer of electroCore.
“Our partnership gives electroCore access to a variety of
contracting portals enabling easier procurement of gammaCore
therapy across veteran and active military channels. We have just
begun to scratch the surface of the help we can provide to our
veterans, troops, and family members in VA and DoD, and we are
honored to partner with an SDVOSB to make gammaCore more widely
available to those veterans and active-duty military who can
benefit from our therapy.”
About Red One Medical Devices, LLCRed One is
transforming healthcare for America’s military service members and
veterans. Red One identifies and delivers advanced medical and
pharmaceutical technologies to the federal government. These
products meet the unique needs of military surgeons and patients.
They are clinically proven to improve treatment outcomes in both
the DoD and VA. Red One accelerates the procurement process,
oftentimes making products available to the government before
they’re offered in the private sector. Their team is on a mission
to provide veterans with top quality care.
With over 90 combined years of experience in medical sales and
government contracting, Red One provides medical and pharmaceutical
companies access to the federal marketplace. Red One is a
government certified Service-Disabled Veteran Owned Small Business
(SDVOSB) and Disability-Owned Business Enterprise (DOBE). Red One
connects leading medical and pharmaceutical manufacturers to US
Department of Veterans Affairs (VA) and Department of Defense (DoD)
hospitals.
About electroCore, Inc.electroCore, Inc. is a
commercial stage bioelectronic medicine company dedicated to
improving patient outcomes through its non-invasive vagus nerve
stimulation therapy platform, initially focused on the treatment of
multiple conditions in neurology. The company's current indications
are the preventive treatment of cluster headache and migraine and
the acute treatment of migraine and episodic cluster headache.For
more information, visit www.electrocore.com.
About gammaCoreTMgammaCoreTM (nVNS) is the
first non-invasive, hand-held medical therapy applied at the neck
as an adjunctive therapy to treat migraine and cluster headache
through the utilization of a mild electrical stimulation to the
vagus nerve that passes through the skin. Designed as a portable,
easy-to-use technology, gammaCore can be self-administered by
patients, as needed, without the potential side effects associated
with commonly prescribed drugs. When placed on a patient’s neck
over the vagus nerve, gammaCore stimulates the nerve’s afferent
fibers, which may lead to a reduction of pain in patients.
gammaCore (nVNS) is FDA cleared in the United States for
adjunctive use for the preventive treatment of cluster headache in
adult patients, the acute treatment of pain associated with
episodic cluster headache in adult patients, and the acute and
preventive treatment of migraine in adolescent (ages 12 and older)
and adult patients. gammaCore is CE-marked in the European Union
for the acute and/or prophylactic treatment of primary headache
(Migraine, Cluster Headache, Trigeminal Autonomic Cephalalgias and
Hemicrania Continua) and Medication Overuse Headache in adults.
gammaCore is contraindicated for patients if they:
- Have an active implantable medical device, such as a pacemaker,
hearing aid implant, or any implanted electronic device
- Have a metallic device, such as a stent, bone plate, or bone
screw, implanted at or near the neck
- Are using another device at the same time (e.g., TENS Unit,
muscle stimulator) or any portable electronic device (e.g., mobile
phone)
Safety and efficacy of gammaCore have not been evaluated in the
following patients:
- Patients diagnosed with narrowing of the arteries (carotid
atherosclerosis)
- Patients who have had surgery to cut the vagus nerve in the
neck (cervical vagotomy)
- Pediatric patients (less than 12 years)
- Pregnant women
- Patients with clinically significant hypertension, hypotension,
bradycardia, or tachycardia
Please refer to the gammaCore Instructions for Use for all of
the important warnings and precautions before using or prescribing
this product.
Forward-Looking StatementsThis press release
and other written and oral statements made by representatives of
electroCore may contain forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
Such forward-looking statements include, but are not limited to,
statements about electroCore's anticipated benefits of accessing
Red One’s federal marketplace network; its business prospects and
clinical and product development plans; its pipeline or potential
markets for its technologies; the timing, outcome and impact of
regulatory, clinical and commercial developments; the availability
and impact of payer coverage, the potential of nVNS generally and
other statements that are not historical in nature, particularly
those that utilize terminology such as "anticipates," "will,"
"expects," "believes," "intends," other words of similar meaning,
derivations of such words and the use of future dates. Actual
results could differ from those projected in any forward-looking
statements due to numerous factors. Such factors include, among
others, the ability to raise the additional funding needed to
continue to pursue electroCore’s business and product development
plans, the inherent uncertainties associated with developing new
products or technologies, the ability to commercialize gammaCore™,
the potential impact and effects of COVID-19 on the business of
electroCore, electroCore’s results of operations and financial
performance, and any measures electroCore has and may take in
response to COVID-19 and any expectations electroCore may have with
respect thereto, competition in the industry in which electroCore
operates and overall market conditions. Any forward-looking
statements are made as of the date of this press release, and
electroCore assumes no obligation to update the forward-looking
statements or to update the reasons why actual results could differ
from those projected in the forward-looking statements, except as
required by law. Investors should consult all of the information
set forth herein and should also refer to the risk factor
disclosure set forth in the reports and other documents electroCore
files with the SEC available at www.sec.gov.
Investors:
Rich Cockrell
CG Capital
404-736-3838
ecor@cg.capital
or
Media Contact:
Jackie Dorsky
electroCore
908-313-6331
Jackie.dorsky@electrocore.com
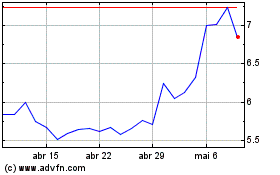
electroCore (NASDAQ:ECOR)
Gráfico Histórico do Ativo
De Jun 2024 até Jul 2024
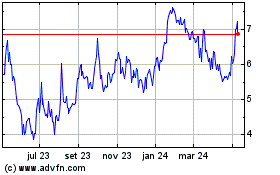
electroCore (NASDAQ:ECOR)
Gráfico Histórico do Ativo
De Jul 2023 até Jul 2024