electroCore Announces Remittance of Annual License Fee from Teijin for Certain Exclusive Rights in Japan
11 Abril 2023 - 9:00AM

electroCore, Inc. (the “Company”) (NASDAQ: ECOR), a
commercial-stage bioelectronic medicine and wellness company, today
announced that Teijin Limited (Teijin), a global conglomerate based
in Japan offering advanced solutions in healthcare, materials and
information technology, has remitted payment of the annual fee
under its license agreement with the Company for certain exclusive
rights to the Company’s non-invasive vagus nerve stimulation (nVNS)
technology for commercialization in Japan for a range of primary
headache disorders.
Under the license agreement, the Company received a
non-refundable, upfront payment for the licenses and rights granted
to Teijin. Teijin’s exclusive rights are subject to a license fee
payable on each anniversary of the license agreement until the
first commercial sale in Japan on any approved indication. By
making the annual license fee payment, Teijin has extended its
exclusive rights for another year.
The financial terms of the license agreement also contain
milestone payments, payable upon the decision by Teijin to
commercialize the licensed product for specific indications. Upon
favorable regulatory and payor coverage decisions in Japan, the
parties will enter into an exclusive commercial supply agreement
for gammaCore™ nVNS.
“Teijin has been an excellent partner thus far as we work
together in navigating the regulatory pathway in Japan,” mentioned
Joshua Lev, Chief Strategy Officer of the Company. “Teijin
continues to show commitment to driving the commercialization of
nVNS in Japan, and we are pleased to work side by side with them as
we advocate for the improvement of the health and lives of primary
headache sufferers across the globe through the use of
nVNS.”
The license agreement contains customary terms and conditions,
including renewal and termination provisions, as well as minimum
purchase commitments once a commercial supply agreement is in
place. Furthermore, Teijin is responsible for all costs associated
with regulatory approval by the Pharmaceuticals and Medical Devices
Agency (PMDA), the Japanese FDA equivalent. As part of the license
agreement, Teijin will have the right of first negotiation for a
license to additional indications in Japan.
About the Teijin GroupTeijin (TSE: 3401) is a
technology-driven global group offering advanced solutions in the
areas of environmental value; safety, security and disaster
mitigation; demographic change and increased health consciousness.
Its main fields of operation are high-performance fibers such as
aramid, carbon fibers & composites, healthcare, films, resin
& plastic processing, polyester fibers, products converting and
IT. The group has over 170 companies and around 20,000 employees
spread out over 20 countries worldwide. It posted consolidated
sales of JPY836.5 billion (USD 6.8 billion) and total assets of JPY
1036.4 billion (USD 8.4 billion) in the fiscal year ending March
31, 2021.
For more information, please visit https://www.teijin.com/.
About electroCore, Inc.electroCore, Inc. is a
commercial stage bioelectronic medicine and wellness company
dedicated to improving health through its non-invasive vagus nerve
stimulation (“nVNS”) technology platform. Our focus is the
commercialization of medical devices for the management and
treatment of certain medical conditions and consumer product
offerings utilizing nVNS to promote general wellbeing and human
performance in the United States and select overseas markets.
For more information, visit www.electrocore.com.
About gammaCore™gammaCore™ (nVNS) is the first
non-invasive, hand-held medical therapy applied at the neck to
treat migraine and cluster headache through the utilization of a
mild electrical stimulation to the vagus nerve that passes through
the skin. Designed as a portable, easy-to-use technology, gammaCore
is self-administered by patients, as needed, without the potential
side effects associated with commonly prescribed drugs. When placed
on a patient’s neck over the vagus nerve, gammaCore stimulates the
nerve’s afferent fibers, which may lead to a reduction of pain in
patients.
gammaCore™ is intended to provide non-invasive vagus nerve
stimulation (nVNS) on the side of the neck for preventive treatment
of migraine and the acute treatment of pain associated with
migraine in patients 12 and older. gammaCore is also indicated for
adjunctive use for preventive treatment of cluster headache, acute
treatment of episodic cluster headache, and treatment of hemicrania
continua and paroxysmal hemicrania in adult patients.
gammaCore should not be used by people with an active
implantable medical device. Please refer to gammaCore.com or the
Instructions for Use for complete safety information, including
indications, contraindications, warnings, cautions, and
instructions.
Forward-Looking StatementsThis press release
and other written and oral statements made by representatives of
electroCore may contain forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
Such forward-looking statements include, but are not limited to,
statements about electroCore's business prospects and clinical and
product development plans (including with respect to enrollment in
ongoing studies); its pipeline or potential markets for its
technologies; the timing, outcome and impact of regulatory,
clinical and commercial developments including online, e-commerce,
direct-to-consumer channels, telehealth portal, and cash pay
initiatives including Japan and its license agreement with Teijin;
the issuance of U.S. and international patents providing expanded
IP coverage; the possibility of future business models and revenue
streams from the Company’s potential use of nVNS for the acute
treatment of PTSD, stroke and hemorrhagic brain injury, the
potential of nVNS generally and gammaCore in particular and other
statements that are not historical in nature, particularly those
that utilize terminology such as "anticipates," "will," "expects,"
"believes," "intends," other words of similar meaning, derivations
of such words and the use of future dates. Actual results could
differ from those projected in any forward-looking statements due
to numerous factors. Such factors include, among others, the
ability to raise the additional funding needed to continue to
pursue electroCore’s business and product development plans, the
inherent uncertainties associated with developing new products or
technologies, the ability to commercialize gammaCore™, the
potential impact and effects of COVID-19 on the business of
electroCore, electroCore’s results of operations and financial
performance, and any measures electroCore has and may take in
response to COVID-19 and any expectations electroCore may have with
respect thereto, competition in the industry in which electroCore
operates and overall market conditions. Any forward-looking
statements are made as of the date of this press release, and
electroCore assumes no obligation to update the forward-looking
statements or to update the reasons why actual results could differ
from those projected in the forward-looking statements, except as
required by law. Investors should consult all of the information
set forth herein and should also refer to the risk factor
disclosure set forth in the reports and other documents electroCore
files with the SEC available at www.sec.gov.
Contact:Rich CockrellCG
Capital404-736-3838ecor@cg.capital
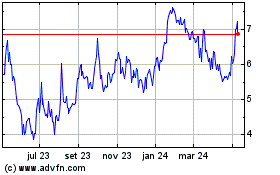
electroCore (NASDAQ:ECOR)
Gráfico Histórico do Ativo
De Jun 2024 até Jul 2024
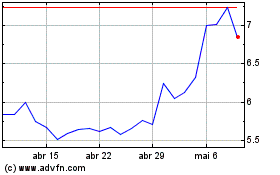
electroCore (NASDAQ:ECOR)
Gráfico Histórico do Ativo
De Jul 2023 até Jul 2024