Hemogenyx Pharmaceuticals and Selexis SA Will Advance Hemogenyx’s Acute Myeloid Leukemia (AML) CDX Bispecific Antibody to Human Trials
12 Janeiro 2022 - 9:00AM
Business Wire
Hemogenyx Pharmaceuticals will leverage
Selexis’ SUREtechnology Platform™ of protein expression
technologies and modular workflows
Hemogenyx Pharmaceuticals plc (LSE: HEMO), the biopharmaceutical
group developing new therapies and treatments for blood diseases,
and Selexis SA, a JSR Life Sciences company, have signed a service
agreement to develop the cell line for Hemogenyx’s CDX bispecific
antibody for the treatment of acute myeloid leukemia (AML). Under
the agreement, Hemogenyx will leverage Selexis’ proprietary
SUREtechnology Platform™, a suite of cell line development tools
and technologies that significantly reduces the time, effort, and
costs associated with developing high-performance mammalian cell
lines.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220112005112/en/
The CDX bispecific is made using the Hemogenyx’s proprietary
humanised monoclonal antibody against a target on the surface of
AML cells. CDX was co-developed by Hemogenyx Pharmaceuticals and
Eli Lilly and Company (“Lilly”). This cutting-edge application of
immune therapy offers a potentially more benign and effective form
of treatment that, if successful, could have a significant impact
on treatment and survival rates for AML. CDX is planned to be the
Company’s second therapeutic candidate to enter clinical trials.
Following the completion of the co-development phase, Lilly granted
the Company an exclusive worldwide license to certain intellectual
property developed by Lilly related to the CDX bispecific antibody
for all uses, including the treatment of AML and other blood
cancers.
Dr. Vladislav Sandler, Chief Executive Officer and co-founder of
Hemogenyx Pharmaceuticals commented, “We are delighted to partner
with Selexis, and access its proprietary protein expression tools
and technologies, IP and know-how. The partnership is key to
advancing our CDX programme into clinical trials and accelerating
the timeline to deliver this innovative therapy to patients in need
of a more benign and effective treatment for AML.”
Mr. Dirk Lange, CEO of Selexis, added, “There’s an urgent need
for effective treatments for AML, and we at Selexis are pleased to
apply our technologies to help Hemogenyx advance the CDX bispecific
antibody to the clinic. We’ve built a reputation for delivering
cell lines rapidly and cost-effectively, without compromising
safety. This is an exciting milestone for Hemogenyx and we welcome
the opportunity to join the company on its journey toward
delivering a promising and effective therapy for patients with
AML.”
Selexis’ modular SUREtechnology Platform™ facilitates the rapid,
stable, and cost-effective production of recombinant proteins and
vaccines, providing seamless integration of the development
continuum from discovery to commercialization.
About Hemogenyx Pharmaceuticals
plc
Hemogenyx Pharmaceuticals is a publicly traded company (LSE:
HEMO) headquartered in London, with its US operating subsidiaries,
Hemogenyx Pharmaceuticals LLC and Immugenyx LLC, located in New
York City at its state-of-the-art research facility.
The Company is a pre-clinical stage biopharmaceutical group
developing new medicines and treatments to treat blood and
autoimmune disease and to bring the curative power of bone marrow
transplantation to a greater number of patients suffering from
otherwise incurable life-threatening diseases. Hemogenyx
Pharmaceuticals is developing several distinct and complementary
product candidates, as well as a platform technology that it uses
as an engine for novel product development.
For more than 50 years, bone marrow transplantation has been
used to save the lives of patients suffering from blood diseases.
The risks of toxicity and death that are associated with bone
marrow transplantation, however, have meant that the procedure is
restricted to use only as a last resort. The Company’s technology
has the potential to enable many more patients suffering from
devastating blood diseases such as leukemia and lymphoma, as well
as severe autoimmune diseases such as multiple sclerosis, aplastic
anemia and systemic lupus erythematosus (Lupus), to benefit from
bone marrow transplantation.
About Selexis SA
Selexis SA, a JSR Life Sciences company, is the global leader in
cell line development with best-in-class modular technology and
highly specialized solutions that enable the life sciences industry
to rapidly discover, develop and commercialize innovative medicines
and vaccines. Our global partners are utilizing Selexis
technologies to advance more than 146 drug candidates in
preclinical and clinical development and the manufacture of eight
commercial products. As part of a comprehensive drug development
process, the Company’s technologies shorten development timelines
and reduce manufacturing risks.
FOR MORE INFORMATION
- Web www.selexis.com - LinkedIn
www.linkedin.com/company/selexis-sa - Twitter
www.twitter.com/SelexisSA - Facebook www.facebook.com/SelexisSA
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220112005112/en/
Enquiries: Hemogenyx Pharmaceuticals plc
https://hemogenyx.com Dr. Vladislav Sandler, Chief Executive
Officer & Co-Founder headquarters@hemogenyx.com Peter Redmond,
Director peter.redmond@hemogenyx.com WuXi Advanced Therapies
Sophie Lutter, Head of Marketing and Communications
atu.info@wuxiapptec.com SP Angel Corporate Finance LLP
Matthew Johnson, Vadim Alexandre, Adam Cowl Tel: +44 (0)20 3470
0470 Peterhouse Capital Limited Lucy Williams, Duncan Vasey,
Charles Goodfellow Tel: +44 (0)20 7469 0930 Selexis
www.selexis.com Company Inquiries Robert Meister, Head,
Corporate Communications robert.meister@selexis.com Tel: +1
602-953-1716 Media Inquiries Mike Beyer mikebeyer@sambrown.com Tel:
+1 312-961-2502
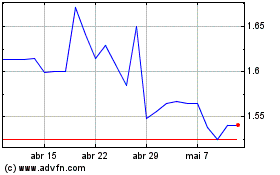
Hemogenyx Pharmaceuticals (LSE:HEMO)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
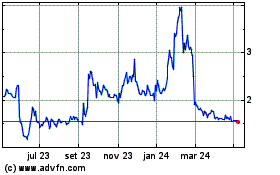
Hemogenyx Pharmaceuticals (LSE:HEMO)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024