VistaGen Therapeutics to Report Fiscal Year 2022 Financial Results on June 23, 2022
17 Junho 2022 - 9:30AM
Business Wire
VistaGen Therapeutics, Inc. (NASDAQ: VTGN) a late
clinical-stage, central nervous system (CNS)-focused
biopharmaceutical company aiming to transform the treatment
landscape for individuals living with anxiety, depression, and
other CNS disorders, today announced it will host a conference call
and webcast on Thursday, June 23, 2022, at 2:00 p.m. Pacific Time
(5:00 p.m. Eastern Time) to report its financial results for fiscal
year 2022 ending on March 31, 2022 and provide a review of recent
achievements and anticipated upcoming milestones.
Event: VistaGen Therapeutics Fiscal Year 2022 Financial
Results Conference Call Date: Thursday, June 23, 2022
Time: 2:00 pm Pacific Time (5:00 pm Eastern Time) US
Dial-in (Toll Free): 1-877-407-9716 TOLL/International
Dial-in: 1-201-493-6779 Conference ID: 13729400
Webcast:
https://viavid.webcasts.com/starthere.jsp?ei=1544875&tp_key=fd5623fb33
An audio webcast of the conference call will also be available
via the link provided above. Participants should access this
webcast site 10 minutes before the start of the call. In addition,
a telephone playback of the call will be available after
approximately 5:00 pm Pacific Time on Thursday, June 23, 2022. To
listen to the replay, call toll free 1-844-512-2921 within the
United States or 1-412-317-6671 when calling internationally
(toll). Please use the replay PIN number 13729400.
About VistaGen VistaGen Therapeutics (Nasdaq: VTGN) is a
late clinical-stage, CNS-focused biopharmaceutical company striving
to transform the treatment landscape for individuals living with
anxiety, depression, and other CNS disorders. The Company is
advancing therapeutics with the potential to be faster-acting, and
with fewer side effects and safety concerns, than those that are
currently available. VistaGen’s clinical-stage candidates are
targeting multiple forms of anxiety and depression. They belong to
a new class of drugs known as pherines, which are odorless,
neuroactive steroids that bind to distinct receptors on
chemosensory neurons in the nasal passages and can impact the
limbic amygdala without systemic uptake or direct activity on CNS
neurons in the brain. VistaGen’s lead candidate, PH94B, is a
nasally administered spray currently in multiple Phase 3 trials in
the U.S., with results anticipated in 2022. Should ongoing Phase 3
studies be successful, PH94B has the potential to be the first
FDA-approved, fast-acting, acute treatment of anxiety for adults
with social anxiety disorder. VistaGen is passionate about
transforming mental health care and redefining what is possible in
the treatment of anxiety and depression. Connect at
www.VistaGen.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220617005073/en/
Investors Mark Flather Vice President, Investor Relations
VistaGen Therapeutics (650) 577-3617 mflather@vistagen.com
Media Nate Hitchings SKDK nhitchings@skdknick.com
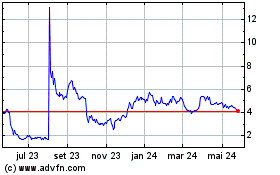
VistaGen Therapeutics (NASDAQ:VTGN)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
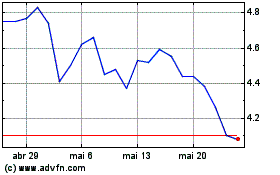
VistaGen Therapeutics (NASDAQ:VTGN)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024