Castle Biosciences to Highlight Clinical Value of IDgenetix® Test at Psych Congress 2023
06 Setembro 2023 - 8:00AM
Business Wire
Castle Biosciences, Inc. (Nasdaq: CSTL), a company improving
health through innovative tests that guide patient care, will share
a poster detailing new data regarding the Company’s IDgenetix® test
at the upcoming Psych Congress 2023 conference, being held in
Nashville, Tennessee, Sept. 6-10. IDgenetix is Castle’s advanced
pharmacogenomic (PGx) test that combines a patient’s drug-drug,
drug-gene and lifestyle-factor interactions into a comprehensive
test report designed to guide medication selection and management
for patients with neuropsychiatric conditions, such as depression
and anxiety.
Details regarding the poster are included below:
- Poster title: The Importance of Incorporating Drug-Drug
Interactions and Lifestyle Factors in Pharmacogenomics-Guided
Medication Management for Patients with Major Depressive Disorder
in a Randomized Controlled Trial
- Poster #: 83
- Date & Time: Friday, Sept. 8, and Saturday, Sept. 9, from
1:30-3 p.m. Central time each day
Posters will remain on display while the exhibit hall is open,
Friday, Sept. 8, and Saturday, Sept. 9, from noon-3:30 p.m. Central
time each day. Additionally, posters will be available for viewing
in the HMP (Healthcare Made Practical) Conferences mobile app and
published on HMP Global’s Psychiatry & Behavioral Health
Learning Network 30-60 days after the event ends.
About IDgenetix®
IDgenetix® is an advanced pharmacogenomic (PGx) test designed to
guide medication selection and management for patients with
neuropsychiatric conditions, such as depression and anxiety.
IDgenetix provides important genetic information to clinicians to
help guide personalized treatment plans for their patients, with a
goal of helping patients achieve faster therapeutic response and
improving their chances of remission by identifying appropriate
medications more efficiently than the standard-of-care,
trial-and-error approach. IDgenetix integrates drug-gene, drug-drug
and lifestyle-factor interactions in a clinically actionable report
and is supported by a published, peer-reviewed randomized
controlled trial that demonstrated clinical utility over the
standard of care when physicians reviewed IDgenetix results prior
to prescribing a medication. More information can be found at
www.IDgenetix.com.
About Castle Biosciences
Castle Biosciences (Nasdaq: CSTL) is a leading diagnostics
company improving health through innovative tests that guide
patient care. The Company aims to transform disease management by
keeping people first: patients, clinicians, employees and
investors.
Castle’s current portfolio consists of tests for skin cancers,
uveal melanoma, Barrett’s esophagus and mental health conditions.
Additionally, the Company has active research and development
programs for tests in other diseases with high clinical need,
including its test in development to predict systemic therapy
response in patients with moderate-to-severe psoriasis, atopic
dermatitis and related conditions. To learn more, please visit
www.CastleBiosciences.com and connect with us on LinkedIn,
Facebook, X and Instagram.
DecisionDx-Melanoma, DecisionDx-CMSeq, DecisionDx-SCC, MyPath
Melanoma, DecisionDx-UM, DecisionDx-PRAME, DecisionDx-UMSeq,
TissueCypher and IDgenetix are trademarks of Castle Biosciences,
Inc.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230906511575/en/
Investor Contact: Camilla Zuckero
czuckero@castlebiosciences.com
Media Contact: Allison Marshall
amarshall@castlebiosciences.com
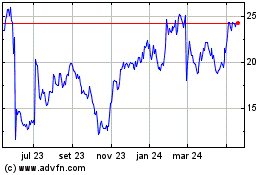
Castle Biosciences (NASDAQ:CSTL)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
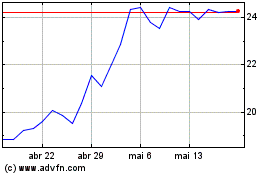
Castle Biosciences (NASDAQ:CSTL)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024