AN2 Therapeutics commences Phase 3 Part of Phase 2/3 Clinical Trial Evaluating Epetraborole for Treatment-Refractory Mycobacterium avium complex (MAC) Lung Disease
13 Setembro 2023 - 8:00AM
Business Wire
Company announces Phase 2 part of the EBO-301 trial is now fully
enrolled with 80 patients; Topline Phase 2 trial results expected
in summer 2024
Over 100 sites across four countries (U.S., Japan, S. Korea
& Australia) as epetraborole enters Phase 3 portion of EBO-301;
expected to enroll ~230 patients
AN2 Therapeutics, Inc. (Nasdaq: ANTX), a clinical-stage
biopharmaceutical company focused on developing treatments for
rare, chronic, and serious infectious diseases with high unmet
needs, today announced that it has commenced the Phase 3 part of
its Phase 2/3 clinical trial evaluating once-daily, oral
epetraborole for treatment-refractory MAC lung disease after
completing Phase 2 enrollment. The Phase 2/3 clinical trial is
expected to support regulatory filings for approval in the U.S. and
Japan. AN2 anticipates reporting Phase 2 topline data in summer
2024.
“Completing Phase 2 enrollment and initiating Phase 3 in this
important clinical trial are significant milestones for AN2 and the
patients we intend to serve. There is currently no approved oral
therapy for MAC lung disease. Given epetraborole’s novel mechanism
of action, encouraging clinical and non-clinical data to date, and
convenient once daily oral dosing, we believe that if approved,
epetraborole has the potential to become the backbone of therapy
for patients suffering with often debilitating treatment-refractory
MAC lung disease,” said Eric Easom, Co-Founder, President and Chief
Executive Officer of AN2 Therapeutics. “We look forward to sharing
topline results from the Phase 2 portion of the trial in summer
2024, at which time we expect to be well on our way to completing
Phase 3 enrollment. We will provide more information on the
projected Phase 3 enrollment timeline as we get closer to the Phase
2 topline readout.”
The FDA has granted epetraborole Fast Track designation for
treatment-refractory MAC lung disease, Qualified Infectious Disease
Product designation for treatment-refractory MAC lung disease, and
orphan drug designation for the treatment of infections caused by
nontuberculous mycobacteria (NTM).
About the Epetraborole Pivotal Phase 2/3 Trial (EBO-301)
This double-blind, placebo-controlled superiority trial uses an
adaptive design to evaluate the safety and efficacy of epetraborole
in patients with treatment-refractory MAC lung disease. The Phase 2
part of the trial (n=80) assesses clinical response using various
patient-reported outcome tools as well as the safety, efficacy and
pharmacokinetics of epetraborole plus an optimized background
regimen (OBR), when compared to a placebo plus an OBR. The Phase 2
findings will be utilized to inform the clinical response measures
evaluated in the Phase 3 part of the trial and confirm the final
sample size. The primary objective in Phase 3 (n≈ 234) will be to
determine if epetraborole plus an OBR, consisting of two or more
standard-of-care drugs, is superior to placebo plus an OBR. For
more information, please visit: www.clinicaltrials.gov
(NCT05327803).
About AN2 Therapeutics
AN2 Therapeutics, Inc. is a clinical-stage biopharmaceutical
company developing treatments for rare, chronic, and serious
infectious diseases with high unmet needs. Our initial candidate is
epetraborole, which we are studying as a once-daily, oral treatment
with a novel mechanism of action for patients with NTM lung
disease, a rare, chronic, and progressive infectious disease caused
by bacteria known as mycobacteria, that leads to irreversible lung
damage and can be fatal. For more information, please visit our
website at www.an2therapeutics.com.
Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995. Forward-looking statements expressed or implied in this press
release include, but are not limited to, statements regarding: the
timing, progress, and anticipated results from AN2's Phase 2/3
pivotal clinical trial; anticipated timing of enrollment in Phase 3
portion of the Phase 2/3 pivotal trial of epetraborole in
treatment-refractory MAC lung disease; anticipated timing of the
data readout of the Phase 2 part of AN2’s pivotal Phase 2/3 trial;
AN2's anticipated progress, business plans, business strategy and
planned clinical trials; the potential clinical benefits and
therapeutic potential of epetraborole; and other statements that
are not historical fact. These statements are based on AN2's
current estimates, expectations, plans, objectives, and intentions,
are not guarantees of future performance and inherently involve
significant risks and uncertainties. Actual results and the timing
of events could differ materially from those anticipated in such
forward-looking statements as a result of these risks and
uncertainties, which include, but are not limited to, risks and
uncertainties related to: the continuing effects of the COVID-19
pandemic; macroeconomic conditions; AN2's ability to progress
enrollment in its Phase 2/3 pivotal clinical trial of epetraborole;
the ability of AN2 to effectively and timely make amendments to the
Phase 2/3 pivotal trial design pursuant to additional FDA feedback;
timely enrollment of patients in its current and future clinical
trials; AN2's ability to procure sufficient supply of its product
candidate for its existing and future clinical trials; the
potential for results from clinical trials to differ from
preclinical, early clinical, preliminary or expected results;
significant adverse events, toxicities or other undesirable side
effects associated with AN2's product candidate; the significant
uncertainty associated with AN2's product candidate ever receiving
any regulatory approvals; AN2's ability to obtain, maintain or
protect intellectual property rights related to its current and
future product candidates; implementation of AN2's strategic plans
for its business and current and future product candidates; the
sufficiency of AN2's capital resources and need for additional
capital to achieve its goals; and other risks, including those
described under the heading “Risk Factors” in AN2's reports to be
filed with the SEC, including AN2’s Report on Form 10-Q for the
quarter ended June 30, 2023. These filings, when available, are
available on the investor relations section of our website at
investor.an2therapeutics.com and on the SEC's website at
www.sec.gov. Forward-looking statements contained in this press
release are made as of this date, and AN2 undertakes no duty to
update such information except as required under applicable
law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230913252729/en/
Lucy O. Day Chief Financial Officer l.day@an2therapeutics.com
Anne Bowdidge Investor Relations abowdidge@an2therapeutics.com
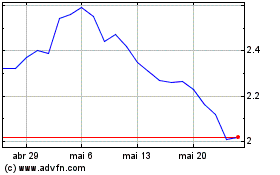
AN2 Therapeutics (NASDAQ:ANTX)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
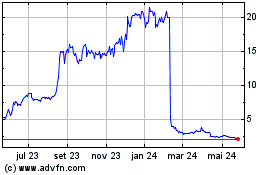
AN2 Therapeutics (NASDAQ:ANTX)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024