Novartis Gets FDA Green Light on Lymphoma Treatment
30 Maio 2022 - 3:09AM
Dow Jones News
By Cecilia Butini
Novartis AG said Saturday that its cancer drug, Kymriah, has
received authorization from the Food and Drug Administration to be
used in the treatment of relapsed or refractory follicular lymphoma
in adults.
The Swiss pharma company said the approval is the third use or
indication of use the drug has received from the FDA for use in
different types of diseases. "In accordance with the Accelerated
Approval Program, continued approval for this indication may be
contingent upon verification and description of clinical benefit in
confirmatory trial(s)," the company said.
Kymriah was already approved in the European Union for relapsed
or refractory follicular lymphoma.
Write to Cecilia Butini at cecilia.butini@wsj.com
(END) Dow Jones Newswires
May 30, 2022 01:54 ET (05:54 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
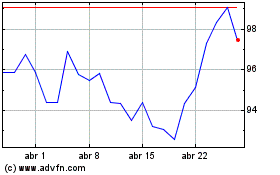
Novartis (NYSE:NVS)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
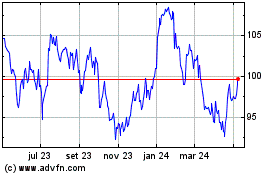
Novartis (NYSE:NVS)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024