Ardelyx Presents Positive Data at DDW 2023 on IBSRELA® (tenapanor), a First-In-Class Treatment for IBS-C in Adults
09 Maio 2023 - 9:00AM

Ardelyx, Inc. (Nasdaq: ARDX), a biopharmaceutical company founded
with a mission to discover, develop and commercialize innovative,
first-in-class medicines that meet significant unmet medical needs,
today announced that a new analysis from IBSRELA Phase 3 trial,
T3MPO-2, was presented in a poster presentation at the 2023
Digestive Disease Week Conference (DDW 2023) that is now underway
in Chicago, Illinois. IBSRELA, discovered and developed by Ardelyx,
is a first-in-class treatment with a novel mechanism and triple
action that is approved by the U.S. Food and Drug Administration to
treat irritable bowel syndrome with constipation (IBS-C) in adults.
“Abdominal symptoms of IBS-C, including pain, bloating and
discomfort, can have an extremely negative impact on patients’
quality of life,” said Brian E. Lacy, M.D., Ph.D., Professor of
Medicine, Mayo Clinic. “The data presented in this poster
demonstrate that patients treated with IBSRELA experienced
significant improvement in abdominal symptoms and that this
improvement correlates with patient reports of adequate relief.
This data analysis provides further evidence of the important role
IBSRELA, with its novel mechanism of action, can play in the
treatment of IBS-C.”
Ardelyx Poster #Tu1618, entitled
“Analysis of Patient-Reported Treatment
Satisfaction and Abdominal Score in Patients with Irritable Bowel
Syndrome with Constipation (IBS-C) with
Tenapanor,” reported results from a post
hoc analysis of the T3MPO-2 study of IBSRELA in adult patients with
IBS-C to examine the relationship between patient-reported
satisfaction (as measured by adequate relief and degree of relief)
and improvement in abdominal symptoms. The Phase 3 T3MPO-2 study
randomized patients to tenapanor 50 mg twice a day or matched
placebo for 26 weeks of treatment. The analysis demonstrated that
IBSRELA meaningfully reduced multiple abdominal symptoms in
patients with IBS-C, including bloating, discomfort and pain,
compared to placebo. The analysis also indicates that
patient-reported treatment satisfaction was strongly correlated
with the IBS-C abdominal score (AS3, combining mean weekly
abdominal pain, bloating and discomfort scores) and could be a
useful tool to assess clinically meaningful improvements in adult
patients with IBS-C.
Poster presentations are now publicly available and can be
accessed on demand HERE.
INDICATION IBSRELA (tenapanor) 50mg BID is
indicated for the treatment of Irritable Bowel Syndrome with
Constipation (IBS-C) in adults.
IMPORTANT SAFETY
INFORMATION
|
WARNING: RISK OF SERIOUS DEHYDRATION IN PEDIATRIC
PATIENTSIBSRELA is contraindicated in patients
less than 6 years of age; in nonclinical studies in young juvenile
rats administration of tenapanor caused deaths presumed to be due
to dehydration. Avoid use of IBSRELA in patients 6 years to less
than 12 years of age. The safety and effectiveness of IBSRELA have
not been established in patients less than 18 years of
age. |
CONTRAINDICATIONS
- IBSRELA is contraindicated in patients less than 6 years of age
due to the risk of serious dehydration.
- IBSRELA is contraindicated in patients with known or suspected
mechanical gastrointestinal obstruction.
WARNINGS AND PRECAUTIONSRisk of Serious
Dehydration in Pediatric Patients
- IBSRELA is contraindicated in patients below 6 years of age.
The safety and effectiveness of IBSRELA in patients less than 18
years of age have not been established. In young juvenile rats
(less than 1 week old; approximate human age equivalent of less
than 2 years of age), decreased body weight and deaths occurred,
presumed to be due to dehydration, following oral administration of
tenapanor. There are no data available in older juvenile rats
(human age equivalent 2 years to less than 12 years).
- Avoid the use of IBSRELA in patients 6 years to less than 12
years of age. Although there are no data in older juvenile rats,
given the deaths in younger rats and the lack of clinical safety
and efficacy data in pediatric patients, avoid the use of IBSRELA
in patients 6 years to less than 12 years of age.
Diarrhea Diarrhea was the most common adverse
reaction in two randomized, double-blind, placebo-controlled trials
of IBS-C. Severe diarrhea was reported in 2.5% of IBSRELA-treated
patients. If severe diarrhea occurs, suspend dosing and rehydrate
patient.
MOST COMMON ADVERSE REACTIONS The most common
adverse reactions in IBSRELA-treated patients (incidence ≥2% and
greater than placebo) were: diarrhea (16% vs 4% placebo), abdominal
distension (3% vs <1%), flatulence (3% vs 1%) and dizziness (2%
vs <1%).
Please see full Prescribing
Information, including Boxed Warning, for
additional risk information.
About Irritable Bowel Syndrome with Constipation
(IBS-C)Irritable bowel syndrome with constipation (IBS-C)
is a gastrointestinal disorder characterized by both abdominal pain
and altered bowel movements, estimated to affect 12 million people
in the U.S. IBS-C is associated with significantly impaired quality
of life, reduced productivity, and substantial economic burden.
About IBSRELA for IBS-C IBSRELA (tenapanor) is
a locally acting inhibitor of the sodium/hydrogen exchanger 3
(NHE3), an antiporter expressed on the apical surface of the small
intestine and colon primarily responsible for the absorption of
dietary sodium. By inhibiting NHE3 on the apical surface of the
enterocytes, tenapanor reduces absorption of sodium from the small
intestine and colon, thus retaining luminal water content, which
accelerates intestinal transit time and results in a softer stool
consistency. IBSRELA has also been shown to reduce abdominal pain
by decreasing visceral hypersensitivity and by decreasing
intestinal permeability in animal models. In a rat model of colonic
hypersensitivity, tenapanor reduced visceral hyperalgesia and
normalized colonic sensory neuronal excitability.
About Ardelyx, Inc.Ardelyx was founded with a
mission to discover, develop and commercialize innovative,
first-in-class medicines that meet significant unmet medical needs.
Ardelyx’s first approved product, IBSRELA® (tenapanor) is available
in the United States and Canada. Ardelyx is developing XPHOZAH®
(tenapanor), a novel product candidate for the control of serum
phosphorus in adult patients with chronic kidney disease (CKD) on
dialysis, which has completed three successful Phase 3 trials.
Ardelyx has a Phase 2 potassium lowering compound, RDX013, for the
potential treatment of elevated serum potassium, or hyperkalemia, a
problem among certain patients with kidney and/or heart disease and
an early-stage program in metabolic acidosis, a serious electrolyte
disorder in patients with CKD. Ardelyx has established agreements
with Kyowa Kirin in Japan, Fosun Pharma in China and Knight
Therapeutics in Canada for the development and commercialization of
tenapanor in their respective territories. For more information,
please visit https://ardelyx.com/ and connect with us on Twitter,
LinkedIn and Facebook.
Investor and Media Contacts: Caitlin
Lowieclowie@ardelyx.com
Kimia Keshtbod kkeshtbod@ardelyx.com
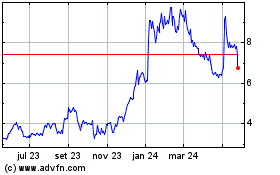
Ardelyx (NASDAQ:ARDX)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
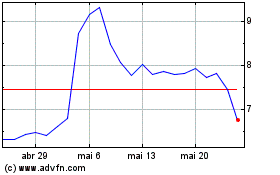
Ardelyx (NASDAQ:ARDX)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024