AIM ImmunoTech Inc. (NYSE American: AIM) (“AIM” or
the “Company”), an immuno-pharma company focused on the research
and development of therapeutics to treat multiple types of cancers,
immune disorders and viral diseases, today provided an update on
its ongoing clinical development programs evaluating Ampligen®
(rintatolimod), a dsRNA and highly selective TLR3 agonist
immune-modulator with broad spectrum activity.
AIM Chief Executive Officer Thomas K. Equels
commented, “We have made promising progress across multiple
clinical fronts, and I am proud of the dedication of our team and
their evident operational execution. Ampligen continues to
demonstrate significant potential across multiple types of cancers,
immune disorders and viral diseases. We are committed to advancing
Ampligen’s development and are poised to achieve multiple key
milestones in 2023.”
Ampligen Phase 2 Study as a Therapy for Locally
Advanced Pancreatic Cancer ("LAPC”) (AMP-270)
The AMP-270 clinical trial is the Company’s
randomized, open-label, controlled, parallel-arm study with the
primary objective of comparing the efficacy of Ampligen versus a no
treatment control group following FOLFIRINOX for subjects with
locally advanced pancreatic adenocarcinoma. Secondary objectives
include comparing safety and tolerability. AMP-270 is expected to
enroll approximately 90 subjects in up to 30 centers across the
United States and Europe.
The Company is recruiting patients for its
AMP-270 Phase 2 study of Ampligen as a therapy for LAPC. The lead
site at the University of Nebraska Medical Center is now open and
actively working to enroll patients. The Gabrail Cancer &
Research Center in Canton, Ohio is also recruiting patients.
Kelsey Klute, MD, Medical Director of the
Pancreatic Diseases Specialty Clinic at Nebraska Medicine,
commented, “We are pleased to join into what we believe is an
important Phase 2 study that has the potential to address a
much-needed treatment option for LAPC. Our team is actively working
to enroll and treat patients on the study in a joint effort with
all other clinical trial sites. We are encouraged by the potential
of Ampligen and look forward to further exploring its potential in
the treatment of LAPC.”
AIM continues its efforts to open additional
clinical sites at premier cancer centers across the United States
and Europe. The Company remains optimistic for first patient
enrollment in the second quarter of this year.
For more information about the AMP-270 please
visit ClinicalTrials.gov and reference identifier NCT05494697.
Ampligen Phase 1 Study for the Treatment of
Early-Stage Triple Negative Breast Cancer (“TNBC”)
Ampligen is currently being evaluated in a Phase
1 study for TNBC by Roswell Park Comprehensive Cancer Center. The
study is designed to evaluate the safety and tolerability of a
combination of Ampligen and celecoxib with or without Intron A,
when given along with chemotherapy. The primary endpoint was safety
and tolerability. Secondary endpoints included pCR rate. Tumor and
blood biomarkers were analyzed in exploratory studies. The goal of
this approach is to increase survival. In November 2022, Roswell
presented positive data at the SITC 37th Annual Meeting
demonstrating the chemokine-modulating regimen including Ampligen
was well tolerated, with promising clinical activity of pathologic
complete response (pCR) + microinvasive residual disease
(ypTmic).
Roswell has now completed the study and is
currently analyzing data. Based on current timelines, the Company
anticipates Roswell will release — via publications — full study
results before the end of the year.
For more information about the Phase 1 study of
Ampligen for the treatment of TNBC, visit ClinicalTrials.gov and
reference identified NCT04081389.
Ampligen Analysis Demonstrating Potential as a
Maintenance Therapy After Systemic Chemotherapy in Patients with
Metastatic and LAPC
A new analysis was recently presented in an
abstract titled, “Immune Response in Stable Pancreatic Cancer after
Rintatolimod Treatment,” by Professor C.H.J. van Eijck, MD, PhD of
Erasmus Medical Center at the 2023 Annual Pancreas Club Meeting
held in Chicago, Illinois. Findings from the analysis demonstrated
that treatment of patients with LAPC after FOLFIRINOX with Ampligen
(rintatolimod) may induce markers of dendritic cells and T cells in
a subgroup of patients. The absence of these markers may predict
tumor progression after FOLFIRINOX, providing the future
possibility of identifying pancreatic cancer patients who could
potentially respond to Ampligen.
The results from the new analysis build on the
previously reported peer-reviewed article “Rintatolimod (Ampligen®)
Enhances Numbers of Peripheral B Cells and Is Associated with
Longer Survival in Patients with Locally Advanced and Metastasized
Pancreatic Cancer Pre-Treated with FOLFIRINOX: A Single-Center
Named Patient Program,” published in March 2022 in the journal
Cancers.
Ampligen as a Potential Therapy Against Ebola
Virus Disease (“EVD”)
The South African Patent and Trademark Office
(CIPC) has granted patent No. 2022/01079, titled “Compositions and
Methods Useful for Ebola Virus Infection.” This pioneering patent
recognizes the efficacy of AIM’s tdsRNA drug family, of which
Ampligen is a member, as a potential solution to combat the
devastating impact of Ebola outbreaks. Under the newly granted
patent, a composition comprising tdsRNA may be used in a method
that involves administering the composition to a subject to
prevent, treat, inhibit, or attenuate an Ebola virus infection in
that subject. Significantly, the composition may be used as a
treatment for individuals already infected with the Ebola virus, or
the composition may be used to prevent an Ebola infection.
About Ampligen
Ampligen is AIM’s dsRNA product candidate being
developed for globally important cancers, viral diseases and
disorders of the immune system. Ampligen has demonstrated in the
clinic the potential for standalone efficacy in a number of solid
tumors. Additionally, Ampligen has shown success in increasing
survival rates and efficacy in the treatment of animal tumors when
used in combination with checkpoint blockade therapies.
Ampligen is currently being evaluated as a
combinational therapy for the treatment of a variety of solid tumor
types in multiple clinical trials – both underway and planned – at
major cancer research centers around the country. Ampligen is being
used to treat pancreatic cancer patients in an Early Access Program
approved by the Inspectorate of Healthcare in the Netherlands at
Erasmus Medical Center. Additionally, Ampligen is also approved in
Argentina for the treatment of severe chronic fatigue syndrome and
is currently being evaluated in SARS-CoV-2/COVID-19, myalgic
encephalomyelitis/chronic fatigue syndrome (ME/CFS) and Post COVID
Conditions.
About AIM ImmunoTech Inc.
AIM ImmunoTech Inc. is an immuno-pharma company
focused on the research and development of therapeutics to treat
multiple types of cancers, immune disorders and viral diseases,
including COVID-19. The Company’s lead product is a first-in-class
investigational drug called Ampligen® (rintatolimod), a dsRNA and
highly selective TLR3 agonist immuno-modulator with broad spectrum
activity in clinical trials for globally important cancers, viral
diseases and disorders of the immune system.
For more information, please visit aimimmuno.com
and connect with the Company on Twitter, LinkedIn, and
Facebook.
Cautionary Statement
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995 (the “PSLRA”). Words such as “may,” “will,”
“expect,” “plan,” “anticipate” and similar expressions (as well as
other words or expressions referencing future events or
circumstances) are intended to identify forward-looking statements.
Many of these forward-looking statements involve a number of risks
and uncertainties. Among other things, for those statements, the
Company claims the protection of safe harbor for forward-looking
statements contained in the PSLRA. The Company does not undertake
to update any of these forward-looking statements to reflect events
or circumstances that occur after the date hereof. The information
found on our website is not incorporated by reference into this
press release and is included for reference purposes
only.
Investor Relations Contact
JTC Team, LLC
Jenene Thomas
833-475-8247
AIM@jtcir.com
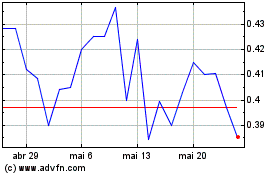
AIM ImmunoTech (AMEX:AIM)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
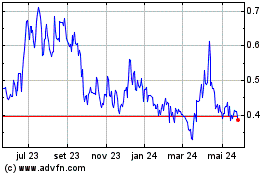
AIM ImmunoTech (AMEX:AIM)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024