INmune Bio Inc. Announces Novel MRI Biomarker Data Demonstrating Improvement in Gray Matter in Patients with Alzheimer’s Disease
11 Julho 2023 - 9:00AM

INmune Bio Inc. (NASDAQ: INMB) (the
“Company”), a clinical-stage immunology company focused on
developing treatments that harness the patient’s innate immune
system to fight disease, reveals new findings from data to be
presented at the annual Alzheimer’s Association International
Conference (AAIC) in Amsterdam, Netherlands. AAIC is the largest
medical meeting focused on Alzheimer’s Disease (AD).
“One of the difficulties in developing drugs for AD is the
inability to quantify the disease and measure changes at the tissue
or microstructure level. The current approaches to measure brain
changes utilize imaging techniques that measure the whole brain,”
said RJ Tesi, M.D., INmune Bio’s Chief Executive Officer. “To
observe whole brain changes in AD often takes a year or longer.
Because whole brain changes are a sum of microstructural
changes, we can more quickly assess the potential of a therapy
through microstructural measurements such as Cortical Disarray
Measurement (CDM®).”
Emerging imaging techniques that assess microstructural changes
are rapidly advancing. INmune Bio is a pioneer in the use of
novel microstructural neuroimaging biomarkers to measure
pharmacodynamic activity of treatment with XProTM in patients
with AD. INmune previously reported that treatment using
XProTM improved microstructural changes in the white
matter tracts that are most affected in AD
patients in participants from our Phase 1b trial. This new
analysis reports similar findings in the gray matter of these same
participants. The results demonstrate a dose dependent enhancement
in gray matter measures throughout the brain in AD patients treated
with XProTM. The changes in microstructural cortical gray matter
structure were measured using Cortical Disarray Measurement and one
metric in particular demonstrated significance PerpPD+. CDM®
is a novel gray matter measure that is more effective than standard
whole brain volumetric changes in predicting cognitive
decline. Notably, the greatest improvement was observed
in the gray matter of brain regions where Alzheimer’s disease
originates.
To better understand the utility
of PerpPD+ in patients with AD treated
with XPro™, associations with other AD biomarkers were assessed.
Baseline levels of PerpPD+ were
highly correlated with baseline levels of cerebral spinal fluid
biomarkers of AD pathology (amyloid and tau), inflammation (YKL-40,
GFAP and sTREM2), and cognitive status (MMSE
scores). “These data provide additional evidence
that XProTM may be having a specific impact on the brain
regions most impacted by AD at the microstructural level within
three months of starting treatment,” said CJ Barnum Ph.D., VP of
CNS Drug Development at INmune Bio. “These findings serve as
indicators of early target engagement for XProTM in AD
and add further support for use of noninvasive diffusion MRI in AD
trials. This new biomarker tool, when added to our existing
suite of biomarkers, should improve the speed, and decrease the
risk of CNS drug development.”
INmune Bio Presentations Discussed at
AAIC 2023
- Cortical
microstructural MRI for detection of early target engagement in AD
drug trials: Post-hoc analysis of exploratory outcomes
from a phase 1b safety study for XPro1595TM in
AD
- Clinical
and biomarker correlates of region-specific diffusion MRI metrics
in a short-term, Phase1b clinical trial for XPro1595TM in
Alzheimer’s disease
About Cortical Disarray Measurement
(CDM®)
PerpPD+ is a novel measure of cortical
diffusivity and key metric included in the Cortical Disarray
Measurement (CDM®) technology developed by Oxford Brain
Diagnostics, Ltd, Oxford (UK). Cortical Disarray Measurement (CDM®)
software has been granted Breakthrough Device designation by the
FDA and is currently in use to assess changes in GM microstructure
as a secondary outcome in our ongoing Phase 2 trial for
XPro1595TM in AD.
The PerpPD+ metric represents the
diffusion components perpendicular to cortical GM minicolumns, is
an indicator of microscopic disruption in cortical GM when
increased in AD and has been shown to be more sensitive than
standard volumetrics to changes in other indicators of synaptic
structure, neuroinflammation and neurodegeneration across the AD
continuum.
About INmune Bio Inc.
INmune Bio Inc.
is a publicly traded (NASDAQ: INMB), clinical-stage
biotechnology company focused on developing treatments that target
the innate immune system to fight disease. INmune Bio has two
product platforms that are both in clinical trials. The DN-TNF
product platform utilizes dominant-negative technology to
selectively neutralize soluble TNF, a key driver of innate immune
dysfunction and mechanistic target of many diseases. DN-TNF is in
clinical trials to determine if it can treat cancer (INB03™), Early
Alzheimer’s disease, and treatment resistant depression (XPro™).
The Natural Killer Cell Priming Platform includes INKmune™ aimed at
priming the patient’s NK cells to eliminate minimal residual
disease in patients with cancer. INmune Bio’s product platforms
utilize a precision medicine approach for the treatment of a wide
variety of hematologic malignancies, solid tumors and chronic
inflammation. To learn more, please
visit www.inmunebio.com.
About Oxford Brain Diagnostics, Ltd.
Oxford Brain Diagnostics Ltd is rethinking how brain health is
assessed and managed. Founded in neuropathological and neuroimaging
expertise, the company’s patented Cortical Disarray Measurement
(CDM®) technology uses MRI brain scan data to support early and
differential diagnosis, track progression, and predict the decline
of neurodegenerative diseases. Oxford Brain Diagnostics is
committed to assessing brain health based on changes in the
cellular structure, supporting drug development, and helping
clinicians around the world in their fight to defeat Alzheimer’s
and other neurodegenerative diseases. For more information,
visit https://www.oxfordbraindiagnostics.com
Forward Looking Statements
Clinical trials are in early stages and there is no assurance
that any specific outcome will be achieved. Any statements
contained in this press release that do not describe historical
facts may constitute forward-looking statements as that term is
defined in the Private Securities Litigation Reform Act of 1995.
Any statements contained in this press release that do not describe
historical facts may constitute forward-looking statements as that
term is defined in the Private Securities Litigation Reform Act of
1995. Any forward-looking statements contained herein are based on
current expectations but are subject to a number of risks and
uncertainties. Actual results and the timing of certain events and
circumstances may differ materially from those described by the
forward-looking statements as a result of these risks and
uncertainties. INB03™, XPro1595™, and INKmune™ are still in
clinical trials or preparing to start clinical trials and have not
been approved by the US Food and Drug Administration (FDA) or any
regulatory body and there cannot be any assurance that they will be
approved by the FDA or any regulatory body or that any specific
results will be achieved. The factors that could cause actual
future results to differ materially from current expectations
include, but are not limited to, risks and uncertainties relating
to the Company’s ability to produce more drug for clinical trials;
the availability of substantial additional funding for the Company
to continue its operations and to conduct research and development,
clinical studies and future product commercialization; and, the
Company’s business, research, product development, regulatory
approval, marketing and distribution plans and strategies. These
and other factors are identified and described in more detail in
the Company’s filings with the Securities and Exchange Commission,
including the Company’s Annual Report on Form 10-K, the Company’s
Quarterly Reports on Form 10-Q and the Company’s Current Reports on
Form 8-K. The Company assumes no obligation to update any
forward-looking statements in order to reflect any event or
circumstance that may arise after the date of this release.
INmune Bio Contact:
David Moss, CFO (858) 964-3720 info@inmunenbio.com
Investor Contact: Jason Nelson Core IR (516)
842-9614 x-823
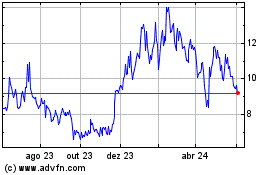
INmune Bio (NASDAQ:INMB)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
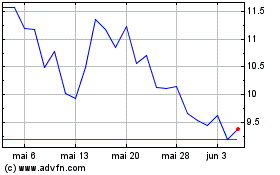
INmune Bio (NASDAQ:INMB)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024