Idorsia presents at the J.P. Morgan Healthcare Conference – Adapting Idorsia for sustainable value creation
10 Janeiro 2024 - 3:00AM

Idorsia presents at the J.P. Morgan Healthcare Conference –
Adapting Idorsia for sustainable value creation
Ad hoc announcement pursuant to Art. 53 LR
- CEO, Jean-Paul Clozel, to update on how Idorsia is being
adapted for sustainable value creation
- The company is focused on extending the cash runway beyond the
current estimate of early April 2024, through various avenues,
including potential out-licensing deals
- Postponement of the publication of Full-Year 2023 Financial
Results and the subsequent Annual General Meeting of
Shareholders
Allschwil, Switzerland – January 10,
2024Idorsia Ltd (SIX: IDIA) today announced that Jean-Paul
Clozel, Chief Executive Officer of Idorsia, will present at the
J.P. Morgan Healthcare Conference on January 10, 2024, at 10:30 PST
/ 19:30 CET. The conference will take place at the Westin St.
Francis hotel in San Francisco, USA.
Jean-Paul will describe how Idorsia is being adapted for
sustainable value creation. The presentation will cover the
progress of QUVIVIQ™ (daridorexant) in the US and Europe, and the
clinical data included in the new drug application for aprocitentan
with the US Food and Drug Administration (FDA) which is currently
under review. He will also present other unencumbered assets that
provide the company with strategic flexibility and several avenues
to explore potential fundraising. Follow this link to access the
audio stream and find the presentation available here.
Jean-Paul Clozel MD and Chief Executive Officer of
Idorsia commented: “Creating a sustainable pharma company
requires scientific innovation and substantial investment. We have
demonstrated our ability to innovate and bring new drugs to the
market and have a portfolio of innovative products, however, we
have limited financing, so we need to prioritize activities that
offer the maximum return in the near term. We estimate the current
cash reserves to last to early April 2024, we therefore plan to
extend the cash runway through various avenues, including potential
out-license deals.”
Expected highlights in 2024
- Secure additional funding to extend the cash runway
- Expand access and availability of QUVIVIQ (daridorexant) in the
US, Canada and across Europe
- Achieve FDA decision for the NDA for aprocitentan in resistant
hypertension
- Achieve EMA decision for the MAA for aprocitentan in resistant
hypertension
Jean-Paul commented on the portfolio: “More
than 11 million QUVIVIQ tablets have been dispensed to help
patients achieve better nights and days. We have made great
progress with access and availability in our key markets, and I
expect to see that translating into income in 2024. I also expect
aprocitentan – the first antihypertensive working on a new pathway
for 30 years – to become available for treated patients whose
hypertension remains uncontrolled. Discussions with health
authorities are going well and I’m confident that the label will
reflect the outstanding results we’ve seen for these high-risk
patients. We also intend to progress the two global Phase 3
programs with selatogrel and cenerimod which have the potential to
transform treatment in their target indications.”
Idorsia’s portfolio
|
Product / compound |
Mechanism of action |
Therapeutic area |
Status |
|
QUVIVIQ™ (daridorexant) |
Dual orexin receptor antagonist |
Insomnia |
Commercially available in the US Germany, Italy,
Switzerland, Spain, the UK, and Canada; Approved in the EU; Phase 2
in pediatric insomnia – recruitingPartners:
Mochida & Sosei in Japan, Simcere in China and Hong Kong |
|
Aprocitentan |
Dual endothelin receptor antagonist |
Resistant hypertension |
NDA under review in the US, MAA under review in the EU, other
filings in preparationPartner: Unencumbered |
|
Lucerastat |
Glucosylceramide synthase inhibitor |
Fabry disease |
Phase 3 primary endpoint not met; open-label extension study
ongoingPartner: Unencumbered |
|
Selatogrel |
P2Y12 inhibitor |
Suspected acute myocardial infarction |
Phase 3 recruitingPartner: Unencumbered |
|
Cenerimod |
S1P1 receptor modulator |
Systemic lupus erythematosus |
Phase 3 recruitingPartner: Unencumbered |
|
ACT-1004-1239 |
ACKR3 / CXCR7 antagonist |
Multiple sclerosis and other demyelinating diseases |
Phase 2 in preparationPartner: Unencumbered |
|
Sinbaglustat |
GBA2/GCS inhibitor |
Rare lysosomal storage disorders |
Phase 1 completePartner: Unencumbered |
|
ACT-1014-6470 |
C5aR1 antagonist |
Immune-mediated disorders |
Phase 1Partner: Unencumbered |
|
ACT-777991 |
CXCR3 antagonist |
Recent-onset Type 1 diabetes |
Phase 1Partner: Unencumbered |
|
IDOR-1117-2520 |
Undisclosed |
Immune-mediated disorders |
Phase 1Partner: Unencumbered |
|
IDO-090 |
Synthetic glycan vaccine |
Clostridium difficile infection |
Phase 1 in preparationPartner: Unencumbered |
Neurocrine Biosciences has a global license to develop and
commercialize ACT-709478 (NBI-827104), Idorsia's novel T-type
calcium channel blocker.
On July 20, 2023, Idorsia sold its operating businesses in the
Asia Pacific (ex-China) region to Sosei Heptares, including the
assignment of the license for PIVLAZ (clazosentan) for the Asia
Pacific (ex-China) region. Idorsia retains the rights to
clazosentan in the rest of the world.
Further details including the current status of each project in
our portfolio can be found in our innovation fact sheet.
Upcoming Financial UpdatesThe company has
decided to postpone the publication of Full-Year 2023 Financial
Results and the subsequent Annual General Meeting of
Shareholders.
- Full-Year 2023 and First Quarter 2024 Financial Results
reporting on April 25, 2024
- Annual General Meeting of Shareholders on June 13,
2024
- Half-Year 2024 Financial Results reporting on July 25,
2024
Notes to the editor
About IdorsiaIdorsia Ltd is reaching out for
more – We have more ideas, we see more opportunities and we want to
help more patients. In order to achieve this, we will develop
Idorsia into a leading biopharmaceutical company, with a strong
scientific core.
Headquartered near Basel, Switzerland – a European biotech-hub –
Idorsia is specialized in the discovery, development and
commercialization of small molecules to transform the horizon of
therapeutic options. Idorsia has a 20-year heritage of drug
discovery, a broad portfolio of innovative drugs in the pipeline,
an experienced team of professionals covering all disciplines from
bench to bedside, and commercial operations in Europe and North
America – the ideal constellation for bringing innovative medicines
to patients.
Idorsia was listed on the SIX Swiss Exchange (ticker symbol:
IDIA) in June 2017 and has over 800 highly qualified specialists
dedicated to realizing our ambitious targets.
For further information, please contactAndrew
C. WeissSenior Vice President, Head of Investor Relations &
Corporate CommunicationsIdorsia Pharmaceuticals Ltd,
Hegenheimermattweg 91, CH-4123 Allschwil+41 58 844 10
10investor.relations@idorsia.commedia.relations@idorsia.com
www.idorsia.com
The above information contains certain "forward-looking
statements", relating to the company's business, which can be
identified by the use of forward-looking terminology such as
"estimates", "believes", "expects", "may", "are expected to",
"will", "will continue", "should", "would be", "seeks", "pending"
or "anticipates" or similar expressions, or by discussions of
strategy, plans or intentions. Such statements include descriptions
of the company's investment and research and development programs
and anticipated expenditures in connection therewith, descriptions
of new products expected to be introduced by the company and
anticipated customer demand for such products and products in the
company's existing portfolio. Such statements reflect the current
views of the company with respect to future events and are subject
to certain risks, uncertainties and assumptions. Many factors could
cause the actual results, performance or achievements of the
company to be materially different from any future results,
performances or achievements that may be expressed or implied by
such forward-looking statements. Should one or more of these risks
or uncertainties materialize, or should underlying assumptions
prove incorrect, actual results may vary materially from those
described herein as anticipated, believed, estimated or
expected.
Idorsia (LSE:0RQE)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
Idorsia (LSE:0RQE)
Gráfico Histórico do Ativo
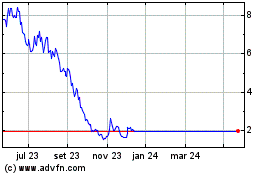
De Mai 2023 até Mai 2024