The Biden-Harris Administration Announces New Guidance to Enable Expanded Access to All FDA-Approved Contraceptives Without Cost
23 Janeiro 2024 - 11:29AM

Agile Therapeutics, Inc., (Nasdaq: AGRX), a women's healthcare
company, commends the Biden-Harris Administration for their
continued commitment to reproductive health including increasing
access to no-cost contraception.
Following several months of focus by the Biden Administration
and lawmakers on contraception and impermissible barriers to
reproductive healthcare, the Departments of Labor, Health and Human
Services, and the Treasury (the “Tri-Agencies”) jointly released
new guidance in the form of Frequently Asked Questions (“FAQ”) on
Monday, January 22, 2024. The FAQ outlines a “new pathway for plans
and issuers to meet existing obligations under federal law by
covering, at no cost, a broader range of FDA-approved contraceptive
drugs and certain devices.”1,2 The Tri-Agencies describe how
plans may comply with the ACA requirement to cover contraception
without cost sharing by “covering all [FDA] approved drugs and
drug-led devices other than those for which there is a covered
therapeutic equivalent” as identified in the Orange Book.1,2 This
new pathway comes after reports of many plans and issuers imposing
impermissible barriers to no-cost contraceptive coverage.
The new guidance follows the President’s Executive Order in June
2023, which directed the Secretaries to consider actions, to the
greatest extent permitted by law, that will ensure coverage of
comprehensive contraceptive care including all contraceptives
approved, granted, or cleared by the Food and Drug Administration,
without cost sharing for enrollees, participants, and
beneficiaries.3
In concert with the FAQ, Secretary Becerra, the head of Health
and Human Services also sent a letter2 to health plans and insurers
highlighting the issuance of new guidance and noting that, “as we
have previously made clear, we will continue to call on group
health plan sponsors and issuers to remove impermissible barriers
and ensure individuals in your plans have access to the
contraceptive coverage they need, as required under the law. It is
more important than ever to ensure access to contraceptive coverage
without cost- sharing, as afforded by the ACA.”
“We recognize the importance of reproductive
freedom for women and applaud the administration for taking
concrete actions to ensure women have access to the contraceptive
product that is right for them without barriers and cost-sharing,”
said Agile Therapeutics, Inc.’s Chairperson and Chief Executive
Officer Al Altomari. “We believe the new guidelines take a
significant step towards eliminating financial barriers to
contraceptives and we look forward to working with patients and
providers interested in a low dose patch option like Twirla®.”
1 HHS Secretary Xavier Becerra Announces New Actions to Increase
Contraceptive Care Coverage on 51st Anniversary of Roe v. Wade HHS
Announces New Actions to Increase Contraceptive Care Coverage
January 22 2024
2 Frequently Asked Questions About the Affordable Care Act Part
64, January 22, 2024 FAQ Part 64 January 22 2024
3 President Biden Executive Order June 23, 2023
Executive-order-on-strengthening-access-to-affordable-high-quality-contraception-and-family-planning-services
June 23, 2023
About Agile
Therapeutics, Inc.Agile
Therapeutics is a women's healthcare company dedicated to
fulfilling the unmet health needs of today’s women. Our product and
product candidates are designed to provide women with contraceptive
options that offer freedom from taking a daily pill, without
committing to a longer-acting method. Our initial product, Twirla®,
(levonorgestrel and ethinyl estradiol), a transdermal system, is a
non-daily prescription contraceptive. Twirla is based on our
proprietary transdermal patch technology, called Skinfusion®, which
is designed to allow drug delivery through the skin. For more
information, please visit the company website at
www.agiletherapeutics.com. The Company may
occasionally disseminate material, nonpublic information on the
Company’s website and LinkedIn account.
About Twirla®Twirla
(levonorgestrel and ethinyl estradiol) transdermal system is a
once-weekly combined hormonal contraceptive (CHC) patch that
contains the active ingredients levonorgestrel (LNG), a type of
progestin, and ethinyl estradiol (EE), a type of estrogen. Twirla
is indicated for use as a method of contraception by women of
reproductive potential with a body mass index (BMI) < 30 kg/m2
for whom a combined hormonal contraceptive is appropriate.
Healthcare providers (HCPs) are encouraged to consider Twirla’s
reduced efficacy in women with a BMI ≥ 25 to <30 kg/m2 before
prescribing. Twirla is contraindicated in women with a BMI ≥ 30
kg/m2. Twirla is also contraindicated in women over 35 years old
who smoke. Cigarette smoking increases the risk of serious
cardiovascular events from CHC use. Twirla is designed to be
applied once weekly for three weeks, followed by a week without a
patch.
Forward-Looking
StatementsCertain information contained in this
press release includes “forward-looking statements”, within the
meaning of Section 27A of the Securities Act of 1933, as amended,
and Section 21E of the Securities Exchange Act of 1934, as amended.
We may, in some cases use terms such as “predicts,” “believes,”
“potential,” “continue,” “anticipates,” “estimates,” “expects,”
“plans,” “intends,” “may,” “could,” “might,” “likely,” “will,”
“should” or other words that convey uncertainty of the future
events or outcomes to identify these forward-looking statements.
Our forward-looking statements are based on current beliefs and
expectations of our management team that involve risks, potential
changes in circumstances, assumptions, and uncertainties, including
statements regarding potential impact of and response to the recent
guidance FAQs issued by the Tri-Agencies including, but not limited
to the potential increase in access to no cost contraception. Any
or all of the forward-looking statements may turn out to be wrong
or be affected by inaccurate assumptions we might make or by known
or unknown risks and uncertainties. These forward-looking
statements are subject to risks and uncertainties including risks
related to the likelihood that the Tri-Agencies will enforce the
new FAQs regarding ACA implementation, the response of commercial
payors to the FAQs and any impact on coverage for Twirla, our
ability to successfully enhance the commercialization of and
increase the uptake for Twirla, the size and growth of the markets
for Twirla and our ability to serve those markets our strategy,
business plans and focus, and the other risks set forth in our
filings with the U.S. Securities and Exchange Commission, including
our Annual Report on Form 10-K and our Quarterly Reports on Form
10-Q. For all these reasons, actual results and developments could
be materially different from those expressed in or implied by our
forward-looking statements. You are cautioned not to place undue
reliance on these forward-looking statements, which are made only
as of the date of this press release. We undertake no obligation to
publicly update such forward-looking statements to reflect
subsequent events or circumstance.
Contact:
Matt Riley
Head of Investor Relations & Corporate Communications
mriley@agiletherapeutics.com
Agile Therapeutics (NASDAQ:AGRX)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
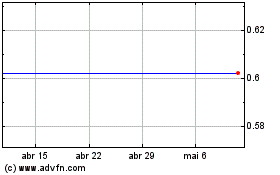
Agile Therapeutics (NASDAQ:AGRX)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024