UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM SD
Specialized
Disclosure Report
AbbVie Inc.
(Exact name of the registrant as specified
in its charter)
Delaware
(State or other Jurisdiction
of
Incorporation) |
001-35565
(Commission File Number) |
32-0375147
(I.R.S. Employer
Identification No.) |
1 North Waukegan Road
North Chicago, Illinois 60064-6400
(Address of principal executive offices) (Zip
code)
Scott T. Reents
(847) 932-7900
(Name and telephone number, including area code,
of the person to contact in connection with this report.)
Check the appropriate box to indicate the rule pursuant to which
this form is being filed, and provide the period to which the information in this form applies:
| |
x |
Rule 13p-1
under the Securities Exchange Act (17 CFR 240.13p-1) for the reporting period from January 1 to December 31, 2023. |
| |
¨ |
Rule 13q-1
under the Securities Exchange Act (17 CFR 240.13q-1) for the fiscal year ended _____. |
Section 1 - Conflict Minerals Disclosure
Item 1.01 Conflict Minerals Disclosure and Report
Conflict Minerals Disclosure
The Conflict Minerals Report for AbbVie Inc. (“AbbVie”)
for the year ended December 31, 2023 is attached as Exhibit 1.01 to this Form SD and is available on AbbVie’s website
at www.abbvieinvestor.com.
The references to AbbVie’s website included in this Form SD
and its exhibits are provided for convenience only, and its contents are not incorporated by reference into this Form SD and Conflict
Minerals Report nor deemed filed with the U.S. Securities and Exchange Commission.
Item 1.02 Exhibit
As specified in this Form SD, AbbVie is hereby filing its Conflict
Minerals Report as Exhibit 1.01.
Section 2 - Resource Extraction Issuer Disclosure
Item 2.01 Resource Extraction Issuer Disclosure and Report
Not applicable.
Section 3 - Exhibits
Item 3.01 Exhibits
The following exhibit is filed as part of this report.
SIGNATURES
Pursuant to the requirements of the Securities
Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the duly authorized undersigned.
| AbbVie Inc. |
|
|
| (Registrant) |
|
|
| |
|
|
|
| By: |
/s/
Scott T. Reents |
|
May 31,
2024 |
|
Scott
T. Reents |
|
(Date) |
| |
Executive Vice President, Chief Financial Officer |
|
|
Exhibit 1.01
AbbVie Inc.
Conflict Minerals Report
For the Year Ended December 31, 2023
As adopted by the Securities
and Exchange Commission (SEC) pursuant to the Dodd-Frank Wall Street Reform and Consumer Protection Act, Rule 13p-1 under the Securities
Exchange Act of 1934 (the Rule) imposes certain reporting obligations on SEC registrants who manufacture, or contract to manufacture,
products for which certain specified minerals are necessary to the functionality or production of the products, regardless of the geographic
origin of the minerals and whether or not they fund armed conflict. These minerals consist of columbite-tantalite (coltan), cassiterite
and wolframite (and their derivatives tantalum, tin, and tungsten), and gold (collectively, 3TG).
This Report covers AbbVie
products manufactured from January 1 through December 31, 2023 for which 3TG may be necessary to the functionality or production.
As used in this Report, the term “AbbVie” refers to AbbVie Inc., a Delaware corporation, and its consolidated subsidiaries.
| |
1. |
Company and Product Overview |
AbbVie is a global, diversified
research-based biopharmaceutical company positioned for success with a comprehensive product portfolio that has leadership positions across
immunology, oncology, aesthetics, neuroscience and eye care. AbbVie uses its expertise, dedicated people and unique approach to innovation
to develop and market advanced therapies that address some of the world’s most complex and serious diseases.
AbbVie has concluded that
certain components for several of its products, such as pump components or electrical components, contain 3TG that is necessary to their
functionality or production (the Products). AbbVie either assembles the Products itself or contracts to have the Products assembled by
a third-party. The Product components are also sourced from third-party suppliers. AbbVie does not directly purchase ore or unrefined
3TG, nor does it have direct relationships with any smelters or refiners. Therefore, AbbVie relies on its direct suppliers to provide
information on the origin of any 3TG contained in the product components they supply to AbbVie, including the source of any 3TG that they
obtain from lower tier suppliers and smelters.
In accordance with the Rule,
AbbVie conducted a good faith reasonable country of origin inquiry (RCOI) that it believes was reasonably designed to determine whether
any of the necessary 3TG contained in the Products originated in the Democratic Republic of the Congo or an adjoining country (Covered
Countries) or from recycled or scrap sources.
As a result of its RCOI, AbbVie
has determined that it has insufficient information to allow it to conclude that (i) it has no reason to believe that any of its
necessary 3TG originated in the Covered Countries or (ii) it reasonably believes its necessary 3TG came from recycled or scrap sources.
AbbVie therefore conducted further due diligence on the source and chain of custody of the necessary 3TG contained in the Products.
Following the exercise of
its due diligence with suppliers, AbbVie is unable to determine at this time the country of origin or mine of origin of the 3TG contained
in the Products. The results of AbbVie’s supply chain due diligence are set forth in this Report, which is published on AbbVie’s
website at www.abbvieinvestor.com.
AbbVie did not purchase 3TG
directly from mines, smelters or refiners. Tracing the minerals to their sources requires AbbVie to rely on its direct suppliers to obtain
information regarding the origin of the 3TG used in the manufacture of the Products. AbbVie’s due diligence measures were designed
to conform in all material respects with the OECD Due Diligence Guidance for Responsible Supply Chains of Minerals from Conflict-Affected
and High-Risk Areas: Third Edition and the related Supplements (OECD Guidance) consistent with its position as a company that is not
involved in the mining, smelting or refining of 3TG. The OECD Guidance is an internationally recognized due diligence framework consisting
of a multi-step, risk-based process. AbbVie has a cross-functional Conflict Minerals Strategy team that is led by Restricted Substances
Management, supervised by the Vice President, Global Directs & Operations and includes stakeholder participation from AbbVie
Finance, Legal, Quality, Procurement, and Commercial functional areas.
AbbVie has established strong
controls to address 3TG. AbbVie’s Supplier Code of Conduct provides that suppliers are expected to ensure that all products and
product components supplied to AbbVie do not contain 3TG that is sourced from Covered Countries and that has directly or indirectly financed
or benefitted armed groups in the Covered Countries. The Supplier Code of Conduct also requires suppliers to have systems in place that
are designed to meet this objective. The Code is published on AbbVie’s website (at https://www.abbvie.com/uploads/PDFs/suppliers/AbbVie-Supplier-Code-of-Conduct-Brochure.pdf).
Violations of the Supplier Code of Conduct, including with respect to 3TG sourcing, are to be reported to AbbVie’s Procurement and
Supplier Management or to the AbbVie Office of Ethics and Compliance. Reports may be made on a confidential and anonymous basis, where
permitted by local law. AbbVie also has a program to monitor supplier social responsibility, which includes surveying certain critical
suppliers on adherence to AbbVie’s Supplier Code of Conduct.
In order to conduct the due
diligence, AbbVie has a process to survey its direct suppliers of the Products, with the objective of providing transparency over the
supply chain and enabling identification of upstream participants such as smelters, together with related controls. AbbVie identified
65 direct suppliers that potentially supply AbbVie with materials containing 3TG for the Products. AbbVie surveyed the majority of these
suppliers, excluding those reporting no 3TG use in previous years or those where AbbVie did not purchase product in 2023. Of those surveyed,
AbbVie requested that these suppliers provide detailed information on a product-level basis. It conducted this survey using the Conflict
Minerals Reporting Template (the Template) developed by the Responsible Minerals Initiative (RMI). The Template is designed to facilitate
a supplier’s disclosure of information regarding 3TG contained in the supplier’s products, including the country of origin
and the name and location of the smelters that process the 3TG. As part of this process, AbbVie also continued to engage with suppliers
concerning their 3TG sourcing and Template responses, including additional communication to address any incomplete or inconsistent information.
At the time of filing AbbVie
did not receive responses from all suppliers surveyed, however, AbbVie is working with the remaining suppliers to complete data collection
efforts. Approximately, 79% of suppliers responded. A portion of the responding suppliers have indicated that their products do not contain
3TG. The suppliers confirming 3TG in their products mostly identified smelters or refiners on a division- or company-wide basis, and,
as a result, not all smelters or refiners identified by these suppliers were necessarily used to process 3TG in the Products.
AbbVie has reviewed the full
smelter and refinery lists provided by the responding suppliers whose products contain 3TG to determine if reported facilities were valid
entities, by comparing those smelting and refining facilities to lists supplied by Trade organizations and Governments. Additionally,
AbbVie reviewed the lists of smelters and refiners determined to be valid entities against lists of smelters and refineries that are certified
to be conflict-free. Lastly, AbbVie researched information regarding each valid smelter or refiner that was not certified as conflict-free
to determine if the facilities are known to source from the Covered Countries. The smelters and refiners that were valid entities and
not certified as conflict-free reportedly sourced minerals from the following countries: Australia, Belgium, Brazil, China, Estonia, France,
Germany, Ghana, India, Indonesia, Italy, Kazakhstan, Republic of Korea, Kyrgyzstan, Lithuania, Malaysia, Mexico, Myanmar,
New Zealand, Norway, Portugal, Russian Federation, Saudi Arabia, South Africa, Sudan, Switzerland, Taiwan Province of China, Thailand,
Turkey, Uganda, United Arab Emirates, United States of America, Vietnam, Zimbabwe.
Nine suppliers had reported
sourcing from a valid smelter that has not been certified as conflict-free and reportedly located in a Covered Country. AbbVie learned
through its own due diligence that this refiner was found to benefit armed conflict. AbbVie continues to work with the suppliers reporting
this refiner to understand the accuracy of the reported refiner within AbbVie’s supply chain. AbbVie requested removal of this refiner
from AbbVie’s supply chain, should the suppliers confirm the accuracy of the reported refiner in AbbVie’s supply chain.
As a result, based on its
reasonable efforts, AbbVie is unable to determine the country of origin, smelter or refiner, or mine of origin of the 3TG specifically
contained in the Products. AbbVie is continuing to work with its suppliers regarding responsible sourcing practices generally and their
3TG supply chain in particular. AbbVie continues to improve its data collection efforts with respect to suppliers and smelters, including
working with suppliers to improve the quality, specificity and reliability of the data they report, such as validity of smelters.
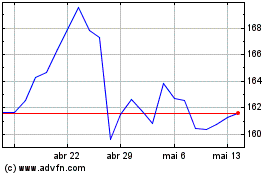
AbbVie (NYSE:ABBV)
Gráfico Histórico do Ativo
De Mai 2024 até Jun 2024
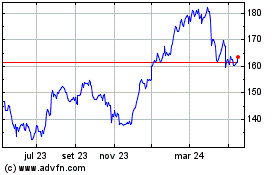
AbbVie (NYSE:ABBV)
Gráfico Histórico do Ativo
De Jun 2023 até Jun 2024