Abbott Gets FDA Approval for Blood Cancer Companion Diagnostic
24 Outubro 2023 - 6:13PM
Dow Jones News
By Ben Glickman
Abbott Laboratories' diagnostic for patients with a rare form of
blood cancer was approved by the U.S. Food and Drug
Administration.
The FDA approved the RealTime IDH1 Assay as a companion
diagnostic for the selection of relapsed or refractory
myelodysplastic syndromes with a mutation in isocitrate
dehydrogenase-1.
The FDA said MDS, a form of blood cancer, can be caused by
mutations in bone marrow cells.
The FDA also approved Tibsovo for the treatment of adult
patients with the same form of MDS, the first targeted therapy
approved for the indication.
Tibsovo was previously approved for some adults with acute
myeloid leukemia and cholangiocarcinoma.
Abbott's test was previously approved as a companion diagnostic
to identify AML patients with the same genetic mutation for
treatment with Tibsovo or Rezlidhia.
Write to Ben Glickman at ben.glickman@wsj.com
(END) Dow Jones Newswires
October 24, 2023 16:58 ET (20:58 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
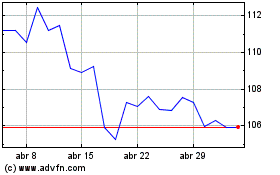
Abbott Laboratories (NYSE:ABT)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
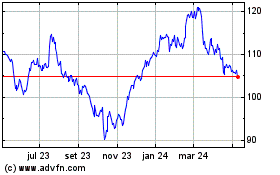
Abbott Laboratories (NYSE:ABT)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024