DARA BioSciences' KRN5500 Granted Orphan Drug Designation by FDA for Treatment of Multiple Myeloma
16 Junho 2014 - 8:00AM
Marketwired Canada
DARA BioSciences, Inc. (NASDAQ: DARA), an oncology supportive care specialty
pharmaceutical company dedicated to providing healthcare professionals a
synergistic portfolio of medicines to help cancer patients adhere to their
therapy and manage side effects arising from their cancer treatments, today
announced the U.S. Food and Drug Administration (FDA) has granted Orphan Drug
Designation to the experimental compound KRN5500 for the treatment of multiple
myeloma.
KRN5500 is a novel, intravenous, non-opioid, non-narcotic compound currently in
Phase 2 clinical development. Earlier this year, KRN5500 received orphan status
to be developed for the parenteral treatment of painful, chronic,
chemotherapy-induced peripheral neuropathy (CCIPN) that is refractory to
conventional analgesics in patients with cancer. In 2011, the FDA designated
KRN5500 as a Fast Track program which expedites the development pathway and
consideration for priority review. Orphan Drug Designation provides DARA with
seven years market exclusivity from the time of approval, tax credits, and the
waiver of PDUFA filing fees, as well as access to federal grants.
"This second orphan drug designation for our key development asset KRN5500
allows us to approach the actual treatment of multiple myeloma, whereas the
CCIPN designation is specific to neuropathy. It is noteworthy in this regard
that up to 20% of myeloma patients have intrinsic peripheral neuropathy, an
incidence that increases to the range of 75% in patients treated with neurotoxic
drugs such as thalidomide or bortezomib. We believe this myeloma-specific orphan
designation enhances both the viability and the future market opportunity for
this valuable pipeline product," said David J. Drutz, M.D., Chief Executive
Officer and Chief Medical Officer of DARA BioSciences.
The favorable consideration of myeloma as an orphan indication for KRN5500 was
supported by a comprehensive publication in 2012 by an independent group of
academic investigators, which demonstrated its therapeutic potential in both in
vitro and in vivo experiments in which KRN5500, a spicamycin derivative,
exhibited anti-myeloma effects through impairing both myeloma cells and
osteoclasts.
Dr. Drutz continued, "We greatly appreciate the expeditious review and approval
of this additional KRN5500 orphan designation application by the FDA's Office of
Orphan Product Development, which provides an important step towards the
potential treatment of multiple myeloma and one of its major complications. This
second orphan drug designation underscores the value of this asset and
strengthens DARA's resolve and positioning in the ongoing pursuit of partnering
opportunities to assist in funding the clinical advancement and development
pathway of KRN5500."
Multiple myeloma is a hematologic cancer (or cancer of the blood) and the second
most common blood cancer after non-Hodgkin's lymphoma. The American Cancer
Society estimates that more than 24,000 new cases of the disease will be
diagnosed in 2014. The FDA grants orphan drug designation to therapeutics
intended to treat diseases that affect fewer than 200,000 people in the United
States.
About DARA BioSciences, Inc.
DARA BioSciences Inc. of Raleigh, North Carolina, is an oncology supportive care
pharmaceutical company dedicated to providing healthcare professionals a
synergistic portfolio of medicines to help cancer patients adhere to their
therapy and manage side effects arising from their cancer treatments.
DARA holds exclusive U.S. marketing rights to both Soltamox® (tamoxifen
citrate) oral solution and Gelclair®. DARA licensed the U.S. rights to
Soltamox® from UK-based Rosemont Pharmaceuticals, Ltd., and Gelclair® from the
Helsinn Group in Switzerland. Under an agreement with Innocutis, DARA also
markets Bionect® (hyaluronic acid sodium salt, 0.2%).
Soltamox® (tamoxifen citrate) oral solution, the only liquid form of tamoxifen,
is indicated for the treatment of metastatic breast cancer, the adjuvant
treatment of node-positive breast cancer in postmenopausal women, the reduction
in risk of invasive breast cancer in women with ductal carcinoma in situ (DCIS),
and for the reduction of the incidence of breast cancer in women at high risk
for breast cancer. Currently, there are more than 1.8 million prescriptions of
tamoxifen written on an annual basis in the United States. Between 30 and 70
percent of patients fail to complete their prescribed course of treatment,
thereby diminishing its benefits in reducing the risk of breast cancer
recurrence.
Tamoxifen Important Safety Information
Tamoxifen citrate is contraindicated in women who require concomitant
coumadin-type anticoagulant therapy, in women with a history of deep vein
thrombosis or pulmonary embolus, and in women with known hypersensitivity to the
drug or any of its ingredients.
Serious and life-threatening events associated with tamoxifen in the risk
reduction setting (women at high risk for cancer and women with DCIS) include
uterine malignancies, stroke and pulmonary embolism.
The most common adverse reactions to tamoxifen treatment are (incidence >
20%) hot flashes, fluid retention, vaginal discharge, vaginal bleeding,
vasodilatation, nausea, irregular menses, weight loss, and musculoskeletal
events.
Tamoxifen carries the following Boxed Warning:
WARNING - For Women with Ductal Carcinoma in Situ (DCIS) and Women at High Risk
for Breast Cancer: Serious and life-threatening events associated with tamoxifen
in the risk reduction setting (women at high risk for cancer and women with
DCIS) include uterine malignancies, stroke and pulmonary embolism. Incidence
rates for these events were estimated from the NSABP P-1 trial (see CLINICAL
PHARMACOLOGY, Clinical Studies, Reduction in Breast Cancer Incidence In High
Risk Women). Uterine malignancies consist of both endometrial adenocarcinoma
(incidence rate per 1,000 women-years of 2.20 for tamoxifen vs. 0.71 for
placebo) and uterine sarcoma (incidence rate per 1,000 women-years of 0.17 for
tamoxifen vs. 0.0 for placebo)*. For stroke, the incidence rate per 1,000
women-years was 1.43 for tamoxifen vs. 1.00 for placebo**. For pulmonary
embolism, the incidence rate per 1,000 women-years was 0.75 for tamoxifen versus
0.25 for placebo**. Some of the strokes, pulmonary emboli, and uterine
malignancies were fatal. Health care providers should discuss the potential
benefits versus the potential risks of these serious events with women at high
risk of breast cancer and women with DCIS considering tamoxifen to reduce their
risk of developing breast cancer. The benefits of tamoxifen outweigh its risks
in women already diagnosed with breast cancer.
*Updated long-term follow-up data (median length of follow-up is 6.9 years) from
NSABP P-1 study. See WARNINGS, Effects on the Uterus-Endometrial Cancer and
Uterine Sarcoma in Prescribing Information. **See Table 3 under CLINICAL
PHARMACOLOGY, Clinical Studies in Prescribing Information.
The full Prescribing Information for Soltamox is available at
www.soltamox.com/prescribing-information.
Gelclair® is an alcohol-free bioadherent oral rinse gel for rapid and effective
relief of pain associated with oral mucositis caused by chemotherapy and
radiation treatment. Gelclair should not be used by patients with a known or
suspected hypersensitivity to the product or any of its ingredients. DARA
licensed the U.S. rights to Soltamox from UK-based Rosemont Pharmaceuticals,
Ltd., and Gelclair from the Helsinn Group in Switzerland. Under an agreement
with Innocutis, DARA also markets Bionect® (hyaluronic acid sodium salt, 0.2%)
a topical treatment for skin irritation and burns associated with radiation
therapy, in U.S. oncology/radiology markets. Bionect should not be used by
patients with known hypersensitivity to any of its ingredients. For further
information on Gelclair and Bionect and the Full Prescribing Information please
visit www.Gelclair.com and www.Bionect.com.
DARA is focused on expanding its portfolio of oncology supportive care products
in the United States, via in-licensing and/or partnering of complementary
late-stage and approved products. In addition, the company wishes to identify a
strategic partner for the clinical development of KRN5500, currently in Phase 2
for the treatment of chronic, treatment refractory, chemotherapy-induced
peripheral neuropathy (CCIPN). The FDA has designated KRN5500 as a Fast Track
Drug, and has granted DARA two separate Orphan Drug Designations for the
treatment of multiple myeloma and for the treatment of painful, chronic
chemotherapy-induced peripheral neuropathy that is refractory to conventional
analgesics (CCIPN).
In early 2014, DARA kicked off its new partnership with Alamo Pharma Services, a
subsidiary of Mission Pharmacal, in deploying a dedicated 20-person national
sales team in the U.S. oncology market. In addition to promoting DARA's products
Soltamox, Gelclair and Bionect, this specialized oncology supportive care sales
team also provides clinicians with access to three Mission Pharmacal products:
Ferralet® 90 (for anemia), BINOSTO® (alendronate sodium effervescent tablet
indicated for the treatment of osteoporosis), and Aquoral® (for
chemotherapy/radiation therapy-induced dry mouth).
Important Safety Information and full Prescribing Information for Mission
Pharmacal's products may be found at: www.Ferralet.com, www.Binosto.com, and
www.Aquoral.com.
For more information please visit our web site at www.darabio.com.
Safe Harbor Statement
All statements in this press release that are not historical are forward-looking
statements within the meaning of the Private Securities Litigation Reform Act of
1995, and are subject to risks and uncertainties. Forward-looking statements are
based on the current expectations, estimates, forecasts and projections
regarding management's beliefs and assumptions. In some cases, you can identify
forward looking statements by terminology such as "may," "will," "should,"
"hope," "expects," "intends," "plans," "anticipates," "contemplates,"
"believes," "estimates," "predicts," "projects," "potential," "continue," and
other similar terminology or the negatives of those terms. Such forward-looking
statements are subject to factors that could cause actual results to differ
materially for DARA from those projected. Important factors that could cause
actual results to differ materially from the expectations described in these
forward-looking statements are set forth under the caption "Risk Factors" in
DARA's most recent Annual Report on Form 10-K, filed with the SEC on February 4,
2014, and DARA's other filings with the SEC from time to time. Those factors
include risks and uncertainties relating to DARA's ability to realize the
desired benefits of Orphan Drug Designation and Fast Track designation for
KRN5500, DARA's ability to timely commercialize and generate revenues or profits
from Soltamox, Gelclair, Bionect or other products given that DARA only recently
hired its initial sales force and DARA's lack of history as a revenue-generating
company, DARA's ability to achieve the desired results from the agreements with
Mission and Alamo, FDA and other regulatory risks relating to DARA's ability to
market Soltamox, Gelclair, Bionect or other products in the United States or
elsewhere, DARA's ability to in-license and/or partner products, DARA's current
cash position and its need to raise additional capital in order to be able to
continue to fund its operations, DARA's ability to raise sufficient capital and
on favorable terms and the stockholder dilution that may result therefrom, the
current regulatory environment in which DARA sells its products, the market
acceptance of those products, dependence on partners, successful performance
under collaborative and other commercial agreements, competition, the strength
of DARA's intellectual property and the intellectual property of others, the
potential delisting of DARA's common stock from the NASDAQ Capital Market, and
other risk factors identified in the documents DARA has filed, or will file,
with the Securities and Exchange Commission ("SEC"). Copies of DARA's filings
with the SEC may be obtained from the SEC Internet site at http://www.sec.gov.
All forward-looking statements are expressly qualified in their entirety by this
cautionary notice. You are cautioned not to place undue reliance on any
forward-looking statements, which speak only as of the date of this release. We
have no obligation, and expressly disclaim any obligation, to update, revise or
correct any of the forward-looking statements, whether as a result of new
information, future events or otherwise.
FOR FURTHER INFORMATION PLEASE CONTACT:
Media Contact:
David Connolly
LaVoieHealthSciences
617-374-8800, Ext. 108
dconnolly@lavoiegroup.com
Corporate Contact:
Jim Polson
FTI Consulting
312 553 6730
Jim.polson@fticonsulting.com
Senvest Capital (TSX:SEC)
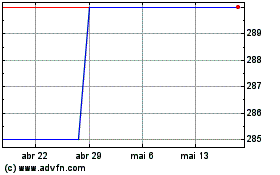
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
Senvest Capital (TSX:SEC)
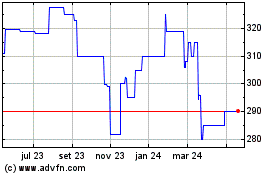
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024