Prothena Announces Appointment of Billy Dunn, M.D., to its Board of Directors
16 Maio 2023 - 5:05PM
Business Wire
Prothena Corporation plc (NASDAQ:PRTA), a late-stage clinical
biotechnology company with a robust pipeline of investigational
therapeutics built on protein dysregulation expertise, announced
today the appointment of Billy Dunn, M.D., to its Board of
Directors. Dr. Dunn is the founding and former Director of the
Office of Neuroscience, Center for Drug Evaluation and Research
(CDER), at the U.S. Food and Drug Administration (FDA).
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20230516005750/en/
Dr. Billy Dunn joins the Prothena Board
of Directors (Photo: Business Wire)
“For nearly two decades, Dr. Dunn has dedicated his career as a
public servant to bringing new and innovative products to patients
suffering from neurodegenerative diseases through his leadership
role at the FDA. His wealth of experience in regulatory and
clinical development, combined with his passion for helping
patients, will greatly benefit the millions that are affected by
diseases caused by protein dysregulation as the company continues
to advance its robust pipeline,” commented Lars G. Ekman, M.D.,
Ph.D., Chair of Prothena’s Board of Directors. “We look forward to
working closely with Dr. Dunn with a shared vision to make
meaningful differences for patients.”
“I am excited to join at this transformational time for
Prothena. The company’s expertise in pioneering therapies targeting
protein dysregulation is clearly reflected in its diverse and
promising pipeline guided by a commitment to patient-focused drug
development, a patient-first approach that I strongly support,”
said Dr. Dunn. “I look forward to working with my fellow board
members and the entire team at Prothena to continue advancing novel
and next-generation medicines for neurodegenerative and rare
peripheral amyloid diseases that have the potential to positively
impact millions of people around the globe. These potential
medicines include an Alzheimer’s disease pipeline with
next-generation anti-amyloid beta and tau-targeted antibodies, and
a vaccine that could be used in prevention paradigms.”
During his tenure at FDA, Dr. Dunn had a significant impact on
how drugs are developed for a range of serious neurological
diseases, including many that previously had only limited treatment
options. His work and leadership contributed to major therapeutic
advances in neuro-immunological diseases such as multiple sclerosis
and myasthenia gravis, advances in rare neurological disorders,
advances in migraine treatment, advances in the range of complex
seizure disorders, and advances in therapies for Alzheimer’s
disease, among many other neurological disorders.
Dr. Dunn is the founding director of the Office of Neuroscience,
CDER, at the FDA, a position he held since the founding of the
office in 2019 through February 2023. He was responsible for the
regulatory oversight of all research conducted to support
neuroscience drug development, including the regulation and review
of investigational new drug applications and marketing applications
for drug and biologic products. From 2005 to 2019, he held
positions of increasing seniority in the Division of Neurology
Products, CDER, including his role as director of that
division.
Dr. Dunn is a trained neurologist and vascular neurologist with
experience in basic research, clinical research, and clinical care.
He earned his B.A. from the University of Virginia and his M.D.
from the F. Edward Hébert School of Medicine in Bethesda,
Maryland.
About Prothena
Prothena Corporation plc is a late-stage clinical biotechnology
company with expertise in protein dysregulation and a pipeline of
investigational therapeutics with the potential to change the
course of devastating neurodegenerative and rare peripheral amyloid
diseases. Fueled by its deep scientific expertise built over
decades of research, Prothena is advancing a pipeline of
therapeutic candidates for a number of indications and novel
targets for which its ability to integrate scientific insights
around neurological dysfunction and the biology of misfolded
proteins can be leveraged. Prothena’s pipeline includes both
wholly-owned and partnered programs being developed for the
potential treatment of diseases including AL amyloidosis, ATTR
amyloidosis, Alzheimer’s disease, Parkinson’s disease and a number
of other neurodegenerative diseases. For more information, please
visit the Company’s website at www.prothena.com and follow the
Company on Twitter @ProthenaCorp.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230516005750/en/
Media Michael Bachner, Senior Director, Corporate Communications
609-664-7308, michael.bachner@prothena.com
Investors IR@prothena.com
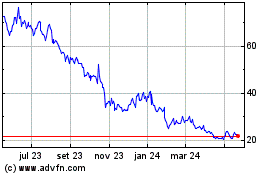
Prothena (NASDAQ:PRTA)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
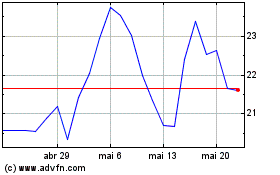
Prothena (NASDAQ:PRTA)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024