FDA Clears AviClear® as a Long-Term Treatment for Mild to Severe Acne
15 Junho 2023 - 9:30AM
Business Wire
AviClear by Cutera® makes history with another
first to market milestone proving to be safe and effective across
all skin types with long lasting results
CUTERA, INC. (Nasdaq: CUTR) a global leader in aesthetic and
dermatology solutions, today announces a new U.S. Food and Drug
Administration Clearance of AviClear as a long-term treatment for mild to severe
inflammatory acne vulgaris. This is the first acne therapy to claim
long term effectiveness for mild, moderate and severe acne.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20230615985076/en/
(Photo: Business Wire)
AviClear initially received FDA clearance in March 2022
following an extensive clinical trial. Now after months of clinical
data evaluation, the FDA has additionally recognized AviClear as a
clinically efficacious and proven treatment for the long-term
treatment of acne. AviClear selectively targets and suppresses the
sebaceous glands, eliminating acne at the source, offering a
durable and prescription free option for patients and
providers.
“Those of us who have been using AviClear on our patients since
the initial FDA Clearance recognized that the results of the
treatment get progressively better with time,” said Emmy M. Graber,
MD, MBA, the Founder of The Dermatology Institute of Boston and an
internationally known acne expert. “I am thrilled that the FDA has
now acknowledged these long-lasting results, giving both patients
and dermatology providers greater confidence in the efficacy and
durability of AviClear results.”
As the first 1726nm laser to be introduced to the market,
AviClear continues to challenge the status quo in the acne
landscape. In three, 30-minute treatment sessions 90% of patients
experienced visible improvement in their acne six months after
their third session.1 According to 12-month clinical data,
improvement increases to 92%2, confirming long-term efficacy of
acne clearance and skin quality over time.
“We are proud to receive such a significant and landmark
designation. The success of AviClear is a testament to Cutera’s
ingenuity and innovation as a pioneering force in results-driven
technology,” said Sheila A. Hopkins, Interim CEO at Cutera.
“Throughout Cutera’s 25-year history, we have continued to develop
devices that offer physicians and their patients breakthrough
treatment options, and AviClear is a great example of our game
changing technologies.”
Interested providers and patients are encouraged to visit
www.AviClear.com for more information.
About Cutera, Inc.
Brisbane, California-based Cutera is a leading provider of
aesthetic and dermatology solutions for practitioners worldwide.
Since 1998, Cutera has been developing innovative, easy-to-use
products that harness the power of science and nature to enable
medical practitioners to offer safe and effective treatments to
their patients. For more information, call +1 415-657-5500 or
1-888-4CUTERA or visit Cutera.com.
1,2 Data on file. FDA clearance study. Cutera, Inc.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230615985076/en/
Media: EvolveMKD – Cutera@EvolveMKD.com Investor Relations: Greg
Barker, VP of FP & A – IR@Cutera.com
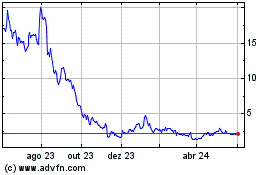
Cutera (NASDAQ:CUTR)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
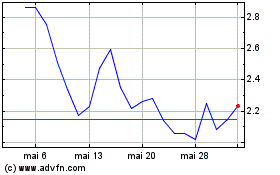
Cutera (NASDAQ:CUTR)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024