Eagle Pharmaceuticals Announces Acceptance of Two Abstracts at the Society for Academic Emergency Medicine (SAEM) Annual Meeting
11 Maio 2023 - 7:50AM

Eagle Pharmaceuticals, Inc. (Nasdaq: EGRX) (“Eagle” or the
“Company”) today announced that two abstracts were accepted for
presentation at the Society for Academic Emergency Medicine (SAEM)
Annual Meeting, being held May 16-19, 2023, in Austin, Texas.
“We are pleased to have the opportunity to present
this investigative work at such a prestigious forum dedicated to
original research in academic emergency medicine. Eagle is actively
engaged with the scientific community, as we focus on developing
innovative medicines to address unmet medical needs. RYANODEX plays
an important role in the treatment of malignant hyperthermia,
today,” stated Scott Tarriff, President and Chief Executive Officer
of Eagle Pharmaceuticals.
Details of the presentations are as follows:
|
Abstract: |
207 |
|
Title: |
Ryanodex-Dantrolene Sodium for Injectable Suspension
Improves Survival in Mouse Model of Acute
Radiation Syndrome |
|
Date: |
Wednesday May 17, 2023 |
|
Time: |
4:16 PM-4:24 PM CDT |
|
Location: |
Room 307: Level Three |
|
|
|
|
Abstract: |
359 |
|
Title: |
Ryanodex® Reduces Persistent Hippocampal Effects of Single
Mild Traumatic Brain Injury in Rats |
|
Date: |
Thursday May 18, 2023 |
|
Time: |
2:38 PM-2:46 PM CDT |
|
Location: |
Room 208: Level Two |
|
|
|
About Eagle Pharmaceuticals, Inc.Eagle is a
fully integrated pharmaceutical company with research and
development, clinical, manufacturing and commercial expertise.
Eagle is committed to developing innovative medicines that result
in meaningful improvements in patients’ lives. Eagle’s
commercialized products include vasopressin, PEMFEXY®, RYANODEX®,
BENDEKA®, BELRAPZO®, TREAKISYM® (Japan), and BYFAVO® and BARHEMSYS®
through its wholly owned subsidiary Acacia Pharma Inc. Eagle’s
oncology and CNS/metabolic critical care pipeline includes product
candidates with the potential to address underserved therapeutic
areas across multiple disease states. Additional information is
available on Eagle’s website at www.eagleus.com.
Forward-Looking Statements This press release
contains “forward-looking statements” within the meaning of the
Private Securities Litigation Reform Act of 1995, as amended, and
other securities law. Forward-looking statements are statements
that are not historical facts. Words and phrases such as
“anticipated,” “forward,” “will,” “would,” “could,” “should,”
“may,” “remain,” “potential,” “prepare,” “expected,” “believe,”
“plan,” “future,” “believe,” “guidance,” “project,” “estimate,”
“intend,” “advance,” “continue” and similar expressions are
intended to identify forward-looking statements. These statements
include, but are not limited to, statements with respect to: the
Company’s ability to develop innovative medicines that address
unmet medical needs; RYANODEX role in the treatment of malignant
hyperthermia; and RYANODEX potential development for other
indications. All of such statements are subject to certain risks
and uncertainties, many of which are difficult to predict and
generally beyond the Company’s control, that could cause actual
results to differ materially from those expressed in, or implied or
projected by, the forward-looking information and statements. Such
risks and uncertainties include, but are not limited to: the risk
that the anticipated benefits of the Company’s acquisition of
Acacia are not realized; the ability of Enalare to achieve
milestones and deliverables and achieve successful results in the
development of ENA-001 and the Company’s ability to exercise its
option to acquire the remaining outstanding share capital of
Enalare; the impacts of the continuing effects of the COVID-19
pandemic and geopolitical events such as the conflict in Ukraine,
including disruption or impact in the sales of the Company's
marketed products, interruptions or other adverse effects to
clinical trials, delays in regulatory review, manufacturing and
supply chain interruptions, adverse effects on healthcare systems,
disruption in the operations of the Company's third party partners
and disruption of the global economy or other events on the
Company's business, financial condition and results of operations;
macroeconomic conditions, including rising inflation and interest
rates, uncertain credit and financial markets and recent and
potential disruptions in banking systems; whether the Company will
incur unforeseen expenses or liabilities or other market factors;
whether the Company will successfully implement its development
plan for its product candidates; delay in or failure to obtain
regulatory approval of the Company's or its partners’ product
candidates; whether the Company can successfully market and
commercialize its product candidates; the success of the Company's
relationships with its partners; the availability and pricing of
third party sourced products and materials; the outcome of
litigation involving any of its products or that may have an impact
on any of our products; successful compliance with the FDA and
other governmental regulations applicable to product approvals,
manufacturing facilities, products and/or businesses; general
economic conditions, including the potential adverse effects of
public health issues, including the COVID-19 pandemic and
geopolitical events, on economic activity and the performance of
the financial markets generally; the strength and enforceability of
the Company's intellectual property rights or the rights of third
parties; competition from other pharmaceutical and biotechnology
companies and the potential for competition from generic entrants
into the market; the risks inherent in the early stages of drug
development and in conducting clinical trials; and any
unanticipated factors in addition to the foregoing that may impact
the Company’s financial and business projections and guidance and
may cause the Company’s actual results and outcomes to materially
differ from its projections and guidance; and those risks and
uncertainties identified in the “Risk Factors” sections of the
Company's Annual Report on Form 10-K for the year ended December
31, 2022, filed with the Securities and Exchange Commission (the
“SEC”) on March 23, 2023, and its other subsequent filings with the
SEC, including the Company’s Quarterly Report on Form 10-Q for the
quarter ended March 31, 2023, filed with the SEC on May 9, 2023.
Readers are cautioned not to place undue reliance on these
forward-looking statements. All forward-looking statements
contained in this press release speak only as of the date on which
they were made. Except to the extent required by law, the Company
undertakes no obligation to update such statements to reflect
events that occur or circumstances that exist after the date on
which they were made.
Investor Relations for Eagle Pharmaceuticals,
Inc.: Lisa M. WilsonIn-Site Communications, Inc. T:
212-452-2793 E: lwilson@insitecony.com
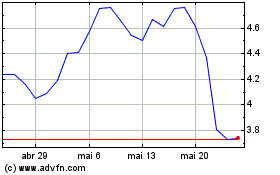
Eagle Pharmaceuticals (NASDAQ:EGRX)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
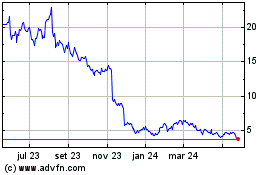
Eagle Pharmaceuticals (NASDAQ:EGRX)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024