Alimera Announces Scientific Data Highlighting ILUVIEN® To Be Presented at the American Academy of Ophthalmology
31 Outubro 2023 - 9:00AM

Alimera Sciences, Inc. (Nasdaq: ALIM) (“Alimera”), a global
pharmaceutical company whose mission is to be invaluable to
patients, physicians and partners concerned with retinal health and
maintaining better vision longer, announces that clinical data for
ILUVIEN® (fluocinolone acetonide intravitreal implant) 0.19 mg
taken from the three-year, phase IV, real-world observational
PALADIN study evaluating the long-term safety of ILUVIEN for
patients with diabetic macular edema, will be featured in three
posters and a presentation on behalf of the PALADIN Investigators,
during the American Academy of Ophthalmology’s 127th Annual Meeting
being held in San Francisco from November 3-6, 2023.
Presentation:Pauline Merrill,
MD “0.19 mg Fluocinolone Acetonide Implant Improves Vision
and Treatment Burden in Patients with DME: The PALADIN Study”
(Sunday November 5, 4:45 p.m. PST)
Posters:David Chin-Yee, MD
“Outcomes in Eyes Requiring IOP Lowering Surgery After Treatment
with the 0.19 Fluocinolone Acetonide Implant for DME: The PALADIN
Study” (Poster Session PO562)
Daniel Kiernan, MD “0.19 mg Fluocinolone
Acetonide Implant Leads to Superior DME Treatment Outcomes in Eyes
with Better Baseline Visual Acuity (≥20/40)” (Poster Session
PO522)
Mathew MacCumber, MD, PhD Control of Retinal
Thickness Variability Improves Vision Following the 0.19mg
Fluocinolone Acetonide Implant in Patients with DME” (Poster
Session PO518)
About ILUVIEN
www.ILUVIEN.com
ILUVIEN (fluocinolone acetonide intravitreal implant) 0.19 mg
sustained release intravitreal implant is injected into the back of
the eye. With its CONTINUOUS MICRODOSING™ technology, ILUVIEN is
designed to release sub-microgram levels of fluocinolone acetonide,
a corticosteroid, for 36 months, to reduce the recurrence of
disease, enabling patients to maintain vision longer with fewer
injections. ILUVIEN is approved in the U.S., Canada, Kuwait,
Lebanon and the U.A.E. to treat diabetic macular edema (DME) in
patients who have been previously treated with a course of
corticosteroids and did not have a clinically significant rise in
intraocular pressure. In 17 European countries, ILUVIEN is
indicated for the treatment of vision impairment associated with
chronic DME considered insufficiently responsive to available
therapies, and for prevention of relapse in recurrent
non-infectious uveitis affecting the posterior segment of the eye.
The 17 European countries include the U.K., Germany, France, Italy,
Spain, Portugal, Ireland, Austria, Belgium, Denmark, Norway,
Finland, Sweden, Poland, Czechia, the Netherlands, and
Luxembourg.
ILUVIEN is marketed for the non-infectious posterior uveitis
indication in Germany, France, the U.K., the Netherlands, Portugal,
Spain, Austria, Ireland, and Italy. ILUVIEN is not approved for
treatment of uveitis in the United States.
About Diabetic Macular Edema (DME)DME, the
primary cause of vision loss associated with diabetic retinopathy,
is a disease affecting the macula, the part of the retina
responsible for central vision. When the blood vessel leakage
associated with diabetic retinopathy results in swelling of the
macula, the condition is called DME. The onset of DME is painless
and may go unreported by the patient until it manifests with the
blurring of central vision or acute vision loss. The severity of
this blurring may range from mild to profound loss of vision. The
Wisconsin Epidemiologic Study of Diabetic Retinopathy found that
over a 10-year period approximately 19% of people with diabetes
included in the study were diagnosed with DME.
About Alimera Sciences,
Inc.www.alimerasciences.comAlimera Sciences is a global
pharmaceutical company whose mission is to be invaluable to
patients, physicians and partners concerned with retinal health and
maintaining better vision longer. For more information, please
visit www.alimerasciences.com.
|
For press inquiries:Jules Abrahamfor Alimera
Sciences 917-885-7378julesa@coreir.com |
For investor inquiries:Scott Gordonfor Alimera
Sciencesscottg@coreir.com |
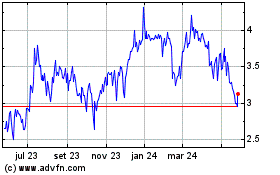
Alimera Sciences (NASDAQ:ALIM)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
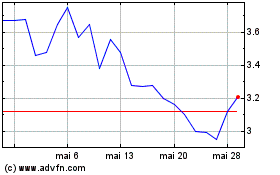
Alimera Sciences (NASDAQ:ALIM)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024