AIM ImmunoTech Completes Treatment of Last Subject in Phase 2 Study Evaluating Ampligen® for the Treatment of Post-COVID Conditions
21 Novembro 2023 - 2:00PM

AIM ImmunoTech Inc. (NYSE American:
AIM) (“AIM” or the “Company”) today announced the
last subject has completed treatment in the Company’s Phase 2 study
evaluating Ampligen® as a potential therapeutic for people with the
Post-COVID condition of fatigue (“AMP-518”).
AIM Chief Executive Officer Thomas K. Equels
commented, “With dosing now complete, our AMP-518 focus turns to
the next milestone of being able to report topline study data as
soon as it is available. We continue to believe in the potential of
Ampligen to provide a much-needed therapeutic option to treat Long
COVID with chronic fatigue-like symptoms. Post-COVID conditions
have emerged as a serious public health threat and a clear public
need.”
As previously announced, the Company completed
enrollment in the AMP-518 study in August 2023. Approximately 80
subjects, ages 18 to 60 years, were enrolled and randomized 1:1 to
receive twice-weekly intravenous infusions of Ampligen or placebo
for 12 weeks, with a follow-up phase of two weeks. To date, no
severe adverse events have been reported. The Company remains on
track to report topline data as early as Q1 2024.
The AMP-518 clinical trial is a two-arm,
randomized, double-blind, placebo-controlled, multicenter study to
evaluate efficacy and safety of Ampligen in subjects experiencing
the post-COVID condition of fatigue. The primary protocol planned
outcome measure of the study is change from baseline to week 13 in
PROMIS® Fatigue Score. Other protocol planned study outcomes
include: change from baseline to week 6 in PROMIS® Fatigue Score;
change from baseline to weeks 6 and 13 in distance traveled during
a Six-Minute Walk Test; proportion of subjects that surpass 54
meters in the Six-Minute Walk Test at the end of 12-week treatment
phase; change from baseline to weeks 6 and 13 in PROMIS® Cognitive
Function Score; and change from baseline to weeks 6 and 13 in
PROMIS® Sleep Disturbance Score.
For more information about AMP-518, please visit
ClinicalTrials.gov and reference identifier NCT05592418.
About AIM ImmunoTech Inc.
AIM ImmunoTech Inc. is an immuno-pharma company
focused on the research and development of therapeutics to treat
multiple types of cancers, immune disorders and viral diseases,
including COVID-19. The company’s lead product is a first-in-class
investigational drug called Ampligen® (rintatolimod), a dsRNA and
highly selective TLR3 agonist immuno-modulator with broad spectrum
activity in clinical trials for globally important cancers, viral
diseases and disorders of the immune system.
For more information, please
visit aimimmuno.com and connect with the Company
on Twitter, LinkedIn, and Facebook.
Cautionary Statement
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995 (the “PSLRA”). Words such as “may,” “will,”
“expect,” “plan,” “anticipate” and similar expressions (as well as
other words or expressions referencing future events or
circumstances) are intended to identify forward-looking statements.
Many of these forward-looking statements involve a number of risks
and uncertainties. Among other things, for those statements, the
Company claims the protection of safe harbor for forward-looking
statements contained in the PSLRA. The Company does not undertake
to update any of these forward-looking statements to reflect events
or circumstances that occur after the date hereof.
Investor Contact:
JTC Team, LLC
Jenene Thomas
(833) 475-8247
AIM@jtcir.com
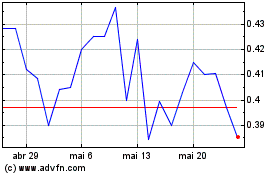
AIM ImmunoTech (AMEX:AIM)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
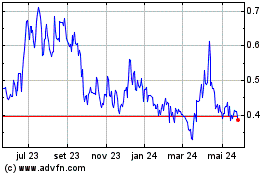
AIM ImmunoTech (AMEX:AIM)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024