Icosavax, Inc. (Nasdaq: ICVX) today announced it has entered into a
definitive agreement pursuant to which AstraZeneca, through an
acquisition subsidiary, will initiate a tender offer to acquire all
of Icosavax’s outstanding shares for a price of $15.00 per share in
cash at closing, plus a non-tradable contingent value right to
receive up to $5.00 in cash, payable upon achievement of specified
regulatory and net sales milestones.
The upfront cash portion of the consideration
represents an equity value of approximately $838 million and a 43%
premium over Icosavax’s closing market price on December 11, 2023,
and a 73% premium to Icosavax’s volume-weighted average price for
the preceding 60 trading days. Combined, the upfront and maximum
potential contingent value payments represent, if achieved, an
equity value of approximately $1.1 billion and a 91% premium over
Icosavax’s closing market price on December 11, 2023, and a 130%
premium to Icosavax’s volume-weighted average price for the
preceding 60 trading days.
The closing of the tender offer is subject to
certain conditions, including the tender of shares representing at
least a majority of the total number of Icosavax’s outstanding
shares, and other customary closing conditions and regulatory
clearances. Upon the successful completion of the tender offer, a
subsidiary of AstraZeneca will be merged with and into Icosavax and
any remaining shares of common stock of Icosavax will be cancelled
and converted into the right to receive the same consideration
(including the contingent value right) per share payable in the
tender offer. Subject to the satisfaction of the conditions in the
merger agreement, the acquisition is expected to close in the first
quarter of 2024.
Adam Simpson, Chief Executive Officer, Icosavax,
said, “We are pleased to announce the proposed acquisition of
Icosavax by AstraZeneca as we believe it offers the opportunity to
accelerate, and expand access to, our potential first-in-class
combination vaccine for older adults at risk from RSV and hMPV. We
look forward to combining our skills and expertise in advancing the
development of IVX-A12, with AstraZeneca’s decades of experience in
RSV, resources, and capabilities in late-stage development.”
Iskra Reic, Executive Vice President, Vaccines
& Immune Therapies, AstraZeneca, said: “This virus-like
particle vaccine technology has the potential to transform
prevention against severe infectious diseases, including RSV and
hMPV. With the addition of Icosavax’s Phase III-ready lead asset to
our late-stage pipeline, we will have a differentiated, advanced
investigational vaccine, and a platform for further development of
combination vaccines against respiratory viruses. This aligns with
our strategy to deliver a portfolio of therapies to address high
unmet needs in infectious diseases, and our ambition to protect the
most vulnerable patients who have high risk of severe
outcomes.”
Concurrent with this press release, Icosavax
issued a press release announcing positive topline interim results
for Icosavax’s Phase 2 study of IVX-A12, a combination virus like
particle (VLP) vaccine candidate targeting both respiratory
syncytial virus (RSV) and human metapneumovirus (hMPV). The press
release can be found at www.icosavax.com.
Centerview Partners LLC is serving as exclusive
financial advisor to Icosavax and Latham & Watkins LLP is
serving as legal counsel.
About Icosavax
Icosavax is a biopharmaceutical company
leveraging its innovative VLP platform technology to develop
vaccines against infectious diseases, with an initial focus on
life-threatening respiratory diseases and a vision for combination
and pan-respiratory vaccines. Icosavax’s VLP platform incorporates
antigen design capabilities and technology to enable multivalent,
particle-based display of complex viral antigens, which it believes
will induce broad, robust, and durable protection against the
specific viruses targeted. Icosavax’s lead program is a combination
vaccine candidate targeting respiratory syncytial virus (RSV) and
human metapneumovirus (hMPV). Its pipeline includes additional
candidates that provide optionality as potential components of
future combination and pan-respiratory vaccines, including
influenza and SARS-CoV-2. Icosavax was formed in 2017 to advance
the breakthrough VLP technology from the Institute for Protein
Design at the University of Washington with the goal to discover,
develop, and commercialize vaccines against infectious diseases.
Icosavax is located in Seattle.
Additional Information and Where to Find
It
The tender offer described above has not yet
commenced. This communication is not an offer to buy nor a
solicitation of an offer to sell any securities of Icosavax, Inc.
The solicitation and the offer to buy shares of Icosavax’s common
stock will only be made pursuant to a tender offer statement on
Schedule TO, including an offer to purchase, a letter of
transmittal and other related materials, that AstraZeneca PLC,
AstraZeneca Finance and Holdings Inc. and Isochrone Merger Sub Inc.
(Merger Sub), a wholly owned indirect subsidiary of AstraZeneca
PLC, intend to file with the Securities and Exchange Commission
(SEC). In addition, Icosavax will file with the SEC a
Solicitation/Recommendation Statement on Schedule 14D-9 with
respect to the tender offer. Once filed, investors will be able to
obtain a free copy of these materials and other documents filed by
AstraZeneca, Merger Sub and Icosavax with the SEC at the website
maintained by the SEC at www.sec.gov. Investors may also obtain, at
no charge, any such documents filed with or furnished to the SEC by
Icosavax under the “Investors & News” section of Icosavax’s
website at www.icosavax.com.
INVESTORS AND SECURITY HOLDERS ARE ADVISED TO
READ THESE DOCUMENTS WHEN THEY BECOME AVAILABLE, INCLUDING THE
SOLICITATION/RECOMMENDATION STATEMENT OF ICOSAVAX AND ANY
AMENDMENTS THERETO, AS WELL AS ANY OTHER DOCUMENTS RELATING TO THE
TENDER OFFER AND THE MERGER THAT ARE FILED WITH THE SEC, CAREFULLY
AND IN THEIR ENTIRETY PRIOR TO MAKING ANY DECISIONS WITH RESPECT TO
WHETHER TO TENDER THEIR SHARES INTO THE TENDER OFFER BECAUSE THEY
CONTAIN IMPORTANT INFORMATION, INCLUDING THE TERMS AND CONDITIONS
OF THE TENDER OFFER.
Forward-Looking Statements
The statements included above that are not a
description of historical facts are forward-looking statements.
Words or phrases such as “believe,” “may,” “could,” “will,”
“estimate,” “continue,” “anticipate,” “intend,” “seek,” “plan,”
“expect,” “should,” “would” or similar expressions are intended to
identify forward-looking statements. The forward-looking statements
are based on the company’s current beliefs and expectations and
include, but are not limited to: statements regarding the planned
completion of the transactions contemplated by the Agreement and
Plan of Merger, dated as of December 11, 2023 (the Merger
Agreement), by and among Icosavax, AstraZeneca and Merger Sub and
the timing thereof; expectations regarding the benefits sought to
be achieved in the transactions; Icosavax’s expectations regarding
the potential benefits and commercial potential of its vaccine
candidates and technology platform; the ability to advance the
company’s development programs and the potential to accelerate and
expand access to IVX-A12 and other future vaccine candidates; and
AstraZeneca’s strategic vision. Risks and uncertainties that could
cause results to differ from expectations include: uncertainties as
to the timing and completion of the tender offer and the merger;
uncertainties as to the percentage of Icosavax stockholders
tendering their shares in the tender offer; the possibility that
competing offers will be made; the possibility that various closing
conditions for the tender offer or the merger may not be satisfied
or waived, including the failure to receive any required regulatory
approvals from any applicable governmental entities (or any
conditions, limitations or restrictions placed on such approvals);
risks that the milestones related to the contingent value rights
are not achieved; the effects of disruption caused by the
transaction making it more difficult to maintain relationships with
employees, collaborators, vendors and other business partners;
risks related to diverting management’s attention from Icosavax’s
ongoing business operations; the risk that stockholder litigation
in connection with the transactions contemplated by the Merger
Agreement may result in significant costs of defense,
indemnification and liability; potential changes in AstraZeneca’s
strategic vision; risks that results of a clinical trial at a
particular time point may not predict future results; potential
delays in the conduct of and receipt of data from clinical trials;
unexpected adverse side effects or inadequate immunogenicity or
efficacy of the company’s vaccine candidates; competing approaches
and approved vaccines limiting the commercial value of the
company’s vaccine candidates; regulatory developments in the United
States and other countries; and other risks and uncertainties
pertaining to Icosavax’s business, including the risks and
uncertainties detailed in Icosavax’s public periodic filings with
the SEC, as well as the tender offer materials to be filed by
AstraZeneca and Merger Sub and the Solicitation/Recommendation
Statement to be filed by Icosavax in connection with the tender
offer.
You are cautioned not to place undue reliance on
these forward-looking statements, which speak only as of the date
hereof. All forward-looking statements are qualified in their
entirety by this cautionary statement and Icosavax undertakes no
obligation to revise or update these statements to reflect events
or circumstances after the date hereof, except as required by
law.
Media Contact:Jessica Yingling,
Ph.D.,Little Dog Communications
Inc.jessica@litldog.com858.344.8091
Investor Contact:Laurence
WattsGilmartin Group, LLClaurence@gilmartinir.com619.916.7620
Icosavax (NASDAQ:ICVX)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
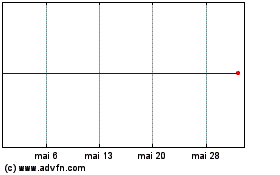
Icosavax (NASDAQ:ICVX)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024