Agilent Companion Diagnostic Expands CE-IVD Mark for PD-L1 IHC 28-8 pharmDx to Include Muscle-Invasive Urothelial Carcinoma (MIUC)
05 Abril 2022 - 9:25AM
Business Wire
Agilent Technologies Inc. (NYSE: A) today announced CE-IVD
marking in the European Union for its PD-L1 IHC 28-8 pharmDx
immunohistochemical assay to extend the use of the test to patients
diagnosed with muscle-invasive urothelial carcinoma (MIUC) who have
tumor cell PD-L1 expression ≥ 1% for adjuvant treatment with
OPDIVO® (nivolumab), a PD-1-targeted immunotherapeutic from Bristol
Myers Squibb.
Urothelial carcinoma is the 10th most common cancer worldwide,
with over 500,000 new cases and approximately 212,000 deaths in
2020.1 The rate of relapse with distant occurrence is approximately
50% in patients with MIUC.2
PD-L1 is a biomarker for response to anti-PD-1 therapies,
including OPDIVO. The expanded use of PD-L1 IHC 28-8 pharmDx helps
physicians in the EU identify MIUC patients for adjuvant treatment
with OPDIVO.
When used in conjunction with PD-L1 IHC 28-8 pharmDx as a
companion test, OPDIVO provides the first and only PD-1-directed
treatment approach that can help reduce the risk of disease
recurrence and offers new hope for patients diagnosed with
MIUC.
“Agilent supports pathologists with products that provide
accuracy and reliability in PD-L1 testing,” said Sam Raha,
president of Agilent's Diagnostics and Genomics Group. “We are
pleased that this now includes the treatment of MIUC for adjuvant
treatment with OPDIVO.”
This new indication CE-marking for PD-L1 IHC 28-8 pharmDx in the
EU expands the current treatment options for urothelial cancer
patients to include MIUC patients whose tumor cells express PD-L1 ≥
1% and are high-risk of recurrence after undergoing radical
resection, for the adjuvant treatment with OPDIVO. The new labeling
builds on Agilent's previous successes in growing the clinical
applicability of therapeutic biomarker analysis, further cementing
the company's position as a provider of diagnostic assays that
deliver high quality and ease of implementation.
References
1.
https://acsjournals.onlinelibrary.wiley.com/doi/full/10.3322/caac.21660
2. John Michael DiBianco, Arvin K George, Daniel Su, Piyush K
Agarwal. Managing noninvasive recurrences after definitive
treatment for muscle-invasive bladder cancer or high-grade upper
tract urothelial carcinoma. Curr Opin Urol. 2015
Sep;25(5):468-75
About Agilent Technologies
Agilent Technologies Inc. (NYSE: A) is a global leader in the
life sciences, diagnostics, and applied chemical markets,
delivering insight and innovation that advance the quality of life.
Agilent’s full range of solutions includes instruments, software,
services, and expertise that provide trusted answers to our
customers' most challenging questions. The company generated
revenue of $6.32 billion in fiscal 2021 and employs 17,000 people
worldwide. Information about Agilent is available at
www.agilent.com. To receive the latest Agilent news, please
subscribe to the Agilent Newsroom. Follow Agilent on LinkedIn,
Twitter, and Facebook.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220405005748/en/
Naomi Goumillout Agilent Technologies +1.781.266.2819
naomi.goumillout@agilent.com
Agilent Technologies (NYSE:A)
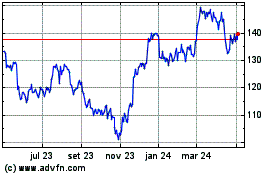
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
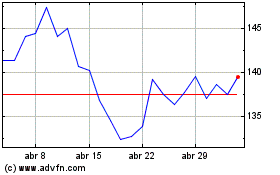
Agilent Technologies (NYSE:A)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024