MedinCell provides additional information regarding the new drug application for MDC-IRM
19 Abril 2022 - 7:34PM
Business Wire
As announced, Teva recently received a Complete Response Letter
(“CRL”) from the U.S. Food and Drug Administration (“FDA”)
regarding the New Drug Application (NDA) for TV-46000/mdc-IRM.
Christophe Douat, CEO of MedinCell, said “We fully trust that
our partner, Teva, will remedy this quickly given the positive
results of the Phase 3 studies. Teva remains confident in
MedinCell’s technology for the development of extended-release
injectable products.”
A Complete Response Letter is issued when the FDA determines
that it cannot approve the application in its current form. Where
possible, the FDA will recommend corrective actions that the
applicant might take to place the application in condition for
approval.
Dr. Richard Malamut, President of MedinCell’s Medical Advisory
Board, said “CRLs are a common part of the FDA regulatory process
and resubmission after addressing identified deficiencies
frequently leads to approval”.
Teva remains committed to the development of extended-release
injectable risperidone and other products based on MedinCell’s
technology.
About MedinCell
MedinCell (Paris:MEDCL) is a pharmaceutical company at
premarketing stage that develops a portfolio of long-acting
injectable products in various therapeutic areas by combining its
proprietary BEPO® technology with active ingredients already known
and marketed. Through the controlled and extended release of the
active pharmaceutical ingredient, MedinCell makes medical
treatments more efficient, particularly thanks to improved
compliance, i.e. compliance with medical prescriptions, and to a
significant reduction in the quantity of medication required as
part of a one-off or chronic treatment. The BEPO® technology makes
it possible to control and guarantee the regular delivery of a drug
at the optimal therapeutic dose for several days, weeks or months
starting from the subcutaneous or local injection of a simple
deposit of a few millimeters, fully bioresorbable. MedinCell
collaborate with tier one pharmaceuticals companies and foundations
to improve Global Health through new therapeutic options. Based in
Montpellier, MedinCell currently employs more than 140 people
representing over 25 different nationalities.
www.medincell.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220419006153/en/
MedinCell David Heuzé Head of communication
david.heuze@medincell.com +33 (0)6 83 25 21 86
NewCap Louis-Victor Delouvrier / Olivier Bricaud Investor
Relations medincell@newcap.eu +33 (0)1 44 71 94 94
NewCap Nicolas Merigeau Media Relations medincell@newcap.eu +33
(0)1 44 71 94 94
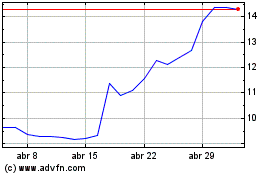
Medincell (EU:MEDCL)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
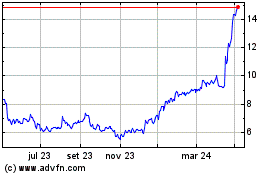
Medincell (EU:MEDCL)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024