Stryker’s Pangea Systems Receive FDA Clearance
12 Setembro 2023 - 10:20AM
Business Wire
Pangea to offer a comprehensive and versatile portfolio,
providing variable-angle plating for a variety of patient
populations
Stryker (NYSE: SYK), one of the world’s leading medical
technology companies, announced that its Pangea Systems including
Femur, Fibula, Tibia, Humerus and Utility have received 510k
clearance from the U.S. Food & Drug Administration.
“FDA clearance is a critical milestone for our Pangea Systems,”
said Eric Tamweber, Vice President and General Manager, Stryker’s
Trauma business unit. “With these new systems, we are now offering
surgeons a comprehensive portfolio that supports a wide range of
their trauma needs.”
Designed by the collaborative efforts of world-renowned
orthopaedic surgeons, the Pangea plates offer an evidence-based
design for implant fit. The system was designed to enhance plate
fit and screw placement while elevating the plating market through
anatomically contoured implants in patient populations with a wide
variety of fracture patterns. The intuitive and streamlined
instrumentation and implant trays will include 20 anatomic plates
and 13 utility plates all accessible in one platform.
The Pangea Systems will be featured at the Annual Orthopaedic
Trauma Association meeting (booth #505) in Seattle on Oct. 18-21.
Attendees will have the opportunity to learn more about the
portfolio and speak with product experts.
About Stryker
Stryker is one of the world’s leading medical technology
companies and, together with its customers, is driven to make
healthcare better. The company offers innovative products and
services in Medical and Surgical, Neurotechnology, Orthopaedics and
Spine that help improve patient and healthcare outcomes. Alongside
its customers around the world, Stryker impacts more than 130
million patients annually. More information is available at
www.stryker.com.
This document is intended solely for healthcare professionals. A
surgeon must always rely on his or her own professional clinical
judgment when deciding whether to use a particular product when
treating a particular patient. Stryker does not dispense medical
advice and recommends that surgeons be trained in the use of any
particular product before using it in surgery.
The information presented is intended to demonstrate the breadth
of Stryker’s product offerings. A surgeon must always refer to the
package insert, product label and/or instructions for use before
using any of Stryker’s products. Products may not be available in
all markets because product availability is subject to the
regulatory and/or medical practices in individual markets. Please
contact your sales representative if you have questions about the
availability of products in your area.
Stryker Corporation or its affiliates own, use, or have applied
for the following trademarks or service marks: Stryker. All other
trademarks are trademarks of their respective owners or
holders.
Content ID: PGA-AR-1, 08-2023
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230912828808/en/
Andrea Sampson President/CEO, Sampson Public Relations Group
asampson@sampsonprgroup.com 562.304.0301
Stryker (NYSE:SYK)
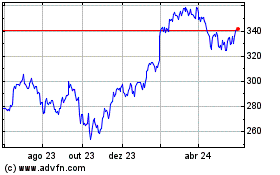
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
Stryker (NYSE:SYK)
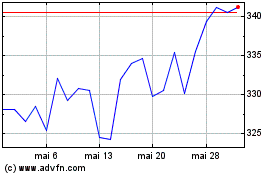
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024