Stryker Launches PROstep® MIS Lapidus, a New Minimally Invasive Surgical Option for Treating Patients with Bunions
19 Setembro 2023 - 10:20AM
Business Wire
Stryker’s PROstep MIS portfolio will be featured at the American
Orthopaedic Foot & Ankle Society Annual Meeting
Stryker (NYSE: SYK), one of the world’s leading medical
technology companies, announced the launch of PROstep® MIS Lapidus,
a new internal fixation system intended for treating bunions using
a minimally invasive surgical reduction of hallux valgus deformity
and subsequent fusion of the first metatarsal cuneiform joint. The
system will debut at the American Orthopaedic Foot & Ankle
Society (AOFAS) Annual Meeting in Louisville, Ky., Sept. 20-23
(booth no. 101).
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20230919818115/en/
Stryker's PROstep® MIS Lapidus is a new
internal fixation system intended for treating bunions using a
minimally invasive surgical reduction of hallux valgus deformity
and subsequent fusion of the first metatarsal cuneiform joint.
(Graphic: Business Wire)
Traditionally, surgeons performing Lapidus bunion procedures use
an open approach, but minimally invasive surgery techniques are
being adopted in numerous surgical specialties because of the
surgeon and patient advantages.1,2 PROstep MIS Lapidus offers
powerful fixation stability through a minimal incision. The new
technology features MIS joint preparation, triplanar reduction, and
a three-screw construct designed to create a tension band for
biomechanical stability.
“This procedure can be a better option for patients who are
worried about cosmetic scarring from surgery and can mitigate the
potential loss of correction caused by improper bone healing,” said
Dr. Alastair Younger, Head of the Distal Extremities Division at
the University of British Columbia. “I appreciate having a viable
minimally invasive surgery option that provides surgeons and their
patients more options for treating bunions.”
Benefits of the PROstep MIS Lapidus system include:
- 2.5% decrease in recurrence compared to open Lapidus
procedures3
- 9% reduction in non-union rates as a result of MIS Lapidus
joint preparation3
- 76% reduction in scar size compared to open bunion correction
procedures4
- Less opioid usage post-op compared to open Lapidus
procedures5
“PROstep MIS Lapidus puts the power back in our surgeon
customers' hands to make the best decision for their patients,”
said Patrick Fisher, Vice President and General Manager for
Stryker’s Foot and Ankle business unit. “At Stryker, we're
continuing to expand our surgery options to support our commitment
to advancing minimally invasive foot and ankle surgery.”
Stryker will host the PROstep MIS Lapidus Lab at AOFAS on Sept.
21 from 12:30-2:00 p.m., in the Stryker mobile lab at the Exhibit
Hall. Design surgeons Dr. Alastair Younger and Dr. Steven Steinlauf
will also be available to meet with attendees during the Meet the
Experts session to demonstrate PROstep MIS Lapidus, at Stryker’s
booth no. 101, on Sept. 20 at 5-6 p.m., Sept. 21 at 10-10:45 a.m.,
and Sept. 22 at 1-2 p.m. ET.
Additionally at AOFAS, Stryker will be debuting the 4WEB Cotton,
Evans, and Utility wedge implants and instruments, which are part
of the 4WEB Osteotomy Truss System™ (OTS). Stryker is now the
exclusive distributor of the OTS that contains the wedge implants
for osteotomies of the foot. OTS has an advanced structural design
that incorporates 4WEB Medical’s proprietary Truss Implant
Technology™.
About Stryker
Stryker is one of the world’s leading medical technology
companies and, together with its customers, is driven to make
healthcare better. The company offers innovative products and
services in Medical and Surgical, Neurotechnology, Orthopaedics and
Spine that help improve patient and healthcare outcomes. Alongside
its customers around the world, Stryker impacts more than 130
million patients annually. More information is available at
www.stryker.com.
Follow Stryker Foot & Ankle on LinkedIn.
A surgeon must always rely on his or her own professional
clinical judgment when deciding whether to use a particular product
when treating a particular patient. Stryker does not dispense
medical advice and recommends that surgeons be trained in the use
of any particular product before using it in surgery.
The information presented is intended to demonstrate the breadth
of Stryker’s product offerings. A surgeon must always refer to the
package insert, product label and/or instructions for use before
using any of Stryker’s products. Products may not be available in
all markets because product availability is subject to the
regulatory and/or medical practices in individual markets. Please
contact your sales representative if you have questions about the
availability of products in your area.
References:
- Trnka HJ, Krenn S, Schuh R. Minimally Invasive Hallux Valgus
Surgery: a Critical Review of the Evidence. International
Orthopaedics 2013; 37: 1731-1735.
- Brogan K, Lindisfarne E, Akehurts H, Farook U, Shrier W, Palmer
S. Minimally Invasive and Open Distal Chevron Osteotomy for Mild to
Moderate Hallux Valgus. Foot Ankle Intl 2016; 37 (11):
1197-1204.
- Diogo, V.C., Velijkovi, A., Wing, K., Penner, M., Gagne, O.,
& Younger, A (2022). Cohort Comparison of Radiographic
Correction and Complications between Minimal Invasive and Open
Lapidus Procedures for Hallux Valgus. Foot & Ankle
International, 43(10), 1277-1284.
https://doi.org/10.1177/10711007221112088.
- Lam P, Lee M, Xing J, & Di Nallo M (2016). Percutaneous
Surgery for Mild to Moderate Hallux Valgus. Foot & Ankle
Clinics N Am, 21(3), 459-477 (data only with respect to chevron
osteotomy procedure)
- Jimmy J. Chan, MD; Javier Z. Guzman, MD; Andrea Nordio, MD;
Jesse C. Chan; Carl M. Cirino, MD; Ettore Vulcano, MD. Opioid
Consumption and Time to Return to Work After Percutaneous Osteotomy
in Foot Surgery. Orthopedics. May 7, 2020.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230919818115/en/
Media contact Andrea Sampson, Sampson Public Relations
Group asampson@sampsonprgroup.com 562.304.0301
Stryker (NYSE:SYK)
Gráfico Histórico do Ativo
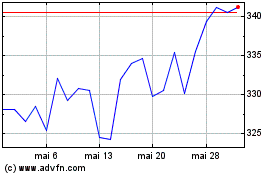
De Abr 2024 até Mai 2024
Stryker (NYSE:SYK)
Gráfico Histórico do Ativo
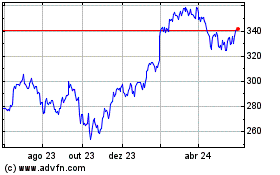
De Mai 2023 até Mai 2024