CARMAT Announces the Opening of Its New Production Facility in Bois-d'Arcy, France
04 Dezembro 2023 - 1:45PM
Business Wire
- The facility and its equipment meet the highest standards for
the production of medical devices
- Its opening will enable the Company to reach a production
capacity of 500 hearts per year as early as 2024, in line with its
objectives
Regulatory News:
CARMAT (FR0010907956, ALCAR), designer and developer of the
world's most advanced total artificial heart, aimed at providing a
therapeutic alternative for patients suffering from advanced
biventricular heart failure (the "Company" or
"CARMAT"), today announced the opening of its second
production facility ("BDA2") in Bois-d'Arcy, in the immediate
vicinity of the Company's first production facility ("BDA1").
The works have now been completed, and the entire facility has
been reviewed by the notified body DEKRA, which in mid-November
2023 approved it for the production of the Aeson® total artificial
heart.
This 1,500 m2 building, which meets the highest medical device
production standards, will enable CARMAT to significantly increase
its capacity in terms of:
- assembly of the electronic parts of the Aeson® heart,
- microbiological testing of the product and the manufacturing
process,
- receiving and checking incoming components,
- storage and shipping.
Combined with the extension of the clean room which is currently
being completed in the "BDA1" facility, the opening of the "BDA2"
facility will enable the Company to reach an annual production
capacity of 500 hearts by early 2024, as planned.
Stéphane Piat, Chief Executive Officer of CARMAT,
comments: "The opening of our new production facility is a major
achievement, and I would like to thank all the teams who have
worked relentlessly to ensure that it is delivered on schedule
before the end of 2023. Today, with the historical "BDA1" facility
and this new "BDA2" facility, we have a high-performance
manufacturing tool that is certified to produce up to 500 hearts
per year from 2024. In line with our strategic plan, we will
further develop our industrial set-up over the next few years, to
reach an annual production capacity of 1,000 Aeson® hearts by
2027."
***
About CARMAT
CARMAT is a French MedTech that designs, manufactures and
markets the Aeson® artificial heart. The Company’s ambition is to
make Aeson® the first alternative to a heart transplant, and thus
provide a therapeutic solution to people suffering from end-stage
biventricular heart failure, who are facing a well-known shortfall
in available human grafts. The world’s first physiological
artificial heart that is highly hemocompatible, pulsatile and
self-regulated, Aeson® could save, every year, the lives of
thousands of patients waiting for a heart transplant. The device
offers patients quality of life and mobility thanks to its
ergonomic and portable external power supply system that is
continuously connected to the implanted prosthesis. Aeson® is
commercially available as a bridge to transplant in the European
Union and other countries that recognize CE marking. Aeson® is also
currently being assessed within the framework of an Early
Feasibility Study (EFS) in the United States. Founded in 2008,
CARMAT is based in the Paris region, with its head offices located
in Vélizy-Villacoublay and its production site in Bois-d’Arcy. The
Company can rely on the talent and expertise of a multidisciplinary
team of circa 200 highly specialized people. CARMAT is listed on
the Euronext Growth market in Paris (Ticker: ALCAR / ISIN code:
FR0010907956).
For more information, please go to www.carmatsa.com and follow
us on LinkedIn.
●●●
Name: CARMAT ISIN code:
FR0010907956 Ticker: ALCAR
●●●
Disclaimer
This press release and the information contained herein do not
constitute an offer to sell or subscribe to, or a solicitation of
an offer to buy or subscribe to, shares in CARMAT (the “Company”)
in any country. This press release may contain forward‐looking
statements that relate to the Company’s objectives and prospects.
Such forward‐looking statements are based solely on the current
expectations and assumptions of the Company’s management and
involve risk and uncertainties including, without limitation, the
Company’s ability to successfully implement its strategy, the rate
of development of CARMAT’s production and sales, the pace and
results of ongoing and future clinical trials, new products or
technological developments introduced by competitors, changes in
regulations and risks associated with growth management. The
Company’s objectives as mentioned in this press release may not be
achieved for any of these reasons or due to other risks and
uncertainties.
The significant and specific risks pertaining to the Company are
those described in the Universal Registration Document (“Document
d’Enregistrement Universel”) filed with the Autorité des Marchés
Financiers (AMF, the French stock market authorities) under number
D. 23-0323. Readers and investors’ attention is, however, drawn to
the fact that other risks, unknown or not deemed to be significant
or specific, may or could exist.
Aeson® is an active implantable medical device commercially
available in the European Union and other countries that recognize
CE marking. The Aeson® total artificial heart is intended to
replace the ventricles of the native heart and is indicated as a
bridge to transplant in patients suffering from end-stage
biventricular heart failure (INTERMACS classes 1-4) who are not
amenable to maximal medical therapy or a left ventricular assist
device (LVAD) and are likely to undergo a heart transplant within
180 days of the device being implanted. The decision to implant and
the surgical procedure must be carried out by healthcare
professionals trained by the manufacturer. The documentation
(clinician manual, patient manual and alarm booklet) should be read
carefully to understand the characteristics of Aeson® and
information necessary for patient selection and the proper use of
Aeson® (contraindications, precautions, side effects). In the
United States, Aeson® is currently exclusively available within the
framework of an Early Feasibility Study authorized by the Food
& Drug Administration (FDA).
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231204535221/en/
CARMAT Stéphane Piat Chief Executive Officer
Pascale d’Arbonneau Chief Financial Officer Tel.: +33 1
39 45 64 50 contact@carmatsas.com
Alize RP Press Relations
Caroline Carmagnol Tel.: +33 6 64 18 99 59
carmat@alizerp.com
NewCap Financial Communication & Investor
Relations
Dusan Oresansky Quentin Massé Tel.: +33 1 44 71 94
92 carmat@newcap.eu
Carmat (EU:ALCAR)
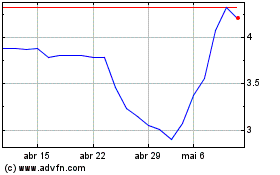
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
Carmat (EU:ALCAR)
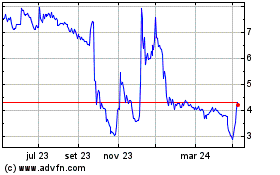
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024