- Abbott’s latest innovation, the FreeStyle Libre® 2 Plus sensor,
is available for the first time for t:slim X2™ insulin pump users
in the U.S.
- Tandem’s Control-IQ™ hybrid closed-loop technology now
integrates with the first and only 15-day continuous glucose
monitoring (CGM) sensor in the U.S.1
Abbott (NYSE: ABT), the global health care company, and Tandem
Diabetes Care, Inc. (NASDAQ: TNDM), a leading insulin delivery and
diabetes technology company, today announced that the t:slim X2
insulin pump with Control-IQ technology is the first automated
insulin delivery (AID) system to integrate with the newly available
FreeStyle Libre 2 Plus sensor, Abbott’s latest continuous glucose
monitoring (CGM) technology. For the first time, FreeStyle Libre
technology users in the United States are now able to experience
the therapeutic benefits of a hybrid closed-loop system that helps
predict and prevent high and low blood sugar.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20240108667517/en/
t:slim X2 Insulin Pump from Tandem
Diabetes Care with Abbott’s Freestyle Libre 2 Plus sensor
integration now available in the United States. (Photo: Business
Wire)
Tandem’s t:slim X2 insulin pump connects wirelessly to the
FreeStyle Libre 2 Plus sensor, which sends automatic glucose
readings every minute2,3 to the pump. Users can conveniently see
their minute-to-minute glucose data on the pump and accompanying
t:connect mobile app.4 The pump’s Control-IQ technology predicts
glucose levels 30 minutes into the future, automatically adjusting
insulin delivery every five minutes based on CGM readings and can
deliver automatic correction boluses (up to one per hour) to help
prevent hyperglycemia.5 When left untreated, hyperglycemia can lead
to long-term diabetes complications, such as nerve and kidney
damage or disease of the eyes.
“Tandem’s leadership in AID innovation is underscored with this
milestone of launching the first insulin pump to be compatible with
Abbott’s CGM technology in the U.S.,” said John Sheridan, president
and chief executive officer of Tandem Diabetes Care. “This
represents another step forward in our commitment to provide
customizable solutions to help reduce burden and create new
possibilities for people living with diabetes.”
Abbott’s latest addition to the FreeStyle Libre portfolio is the
FreeStyle Libre 2 Plus sensor, which is the modified FreeStyle
Libre 2 sensor cleared in 2023 by the U.S. Food & Drug
Administration for use with AID systems. It is the first and only
CGM available in the United States with a wear time of 15 days1 for
both adults and children. Demonstrating proven accuracy in overall
glucose readings with an 8.2% MARD6, the sensor is small, discreet7
and comfortable8 to wear.
“Our FreeStyle Libre portfolio empowers people living with
diabetes to have more control over their health and eases the
burden of their condition by using a continuous glucose monitoring
system with unsurpassed accuracy6 that is simple, easy and
affordable to use,” said Jared Watkin, executive vice president of
Abbott’s diabetes care business. “Through the integration of
Tandem’s t:slim X2 insulin pump with our FreeStyle Libre 2 Plus
sensor, people only need two sensors a month, which ensures both a
more convenient and more affordable experience.”
Tandem will email instructions to all in-warranty t:slim X2
users in the U.S. offering the option to add the new FreeStyle
Libre 2 Plus sensor integration at no cost via remote software
update. t:slim X2 pumps pre-loaded with the updated software are
now shipping to new customers. Information about the software
update process for existing in-warranty pump users, including
system requirements, is available at
tandemdiabetes.com/softwareupdate.
The FreeStyle Libre 2 Plus sensor is available now at
participating durable medical equipment (DME) suppliers. Please
contact Abbott’s Customer Care Team at 1-844-330-5535 for a list of
the participating DMEs.
For more information on how to access the t:slim X2 insulin pump
and FreeStyle Libre 2 Plus sensor integration, please visit:
tandemdiabetes.com/products.
1. Based on patient applied sensors currently on the market in
the US. 2. FreeStyle Libre 2 Plus sensor connects via Bluetooth to
the Tandem t:slim X2 insulin pump. Glucose data is visualized on
the t:connect mobile app and t:slim X2 insulin pump. 3. Not
applicable to insulin delivery adjustment frequency. Refer to
Tandem t:slim X2 pump User Guide for specific details about how the
Control-IQ technology algorithm adjusts insulin delivery. 4. The
information on the t:connect mobile app display may not be
identical to the current status of your pump. Wireless uploads from
the t:connect mobile app to the cloud-based t:connect web
application require a compatible phone and an internet or wireless
data connection. Uploads to the t:connect web application do not
take place in real time and should not be relied upon by healthcare
providers or caregivers for remote patient monitoring. Standard
carrier data rates may apply. 5. If glucose values are predicted to
be above 180 mg/dL, Control-IQ technology calculates a correction
bolus using the Personal Profile settings and a target of 110 mg/dL
and delivers 60% of that value. 6. Data on file, Abbott Diabetes
Care, Inc. FreeStyle Libre 2 User’s Manual 7. Data on file, Abbott
Diabetes Care, Inc. 8. Haak, T. Diabetes Therapy (2017):
https://doi.org/10.1007/s13300-016-0223-6.
About Abbott
Abbott is a global healthcare leader that helps people live more
fully at all stages of life. Our portfolio of life-changing
technologies spans the spectrum of healthcare, with leading
businesses and products in diagnostics, medical devices,
nutritionals and branded generic medicines. Our 115,000 colleagues
serve people in more than 60 countries.
Connect with us at www.abbott.com, on LinkedIn at
www.linkedin.com/company/abbott, on Facebook at
www.facebook.com/Abbott and on Twitter/X @AbbottNews.
About Tandem Diabetes Care
Tandem Diabetes Care, Inc., a global insulin delivery and
diabetes technology company based in San Diego, California, creates
new possibilities for people living with diabetes, their loved
ones, and healthcare providers through a positively different
experience. The Company’s human-centered approach to design,
development, and support delivers innovative products and services
for people who use insulin. Tandem manufactures and sells the
t:slim X2 insulin pump with Control-IQ technology. For more
information, visit tandemdiabetes.com.
Follow Tandem Diabetes Care on Twitter @tandemdiabetes; use
#tslimX2 and #TandemDiabetes. Follow Tandem Diabetes Care on
Facebook at www.facebook.com/TandemDiabetes. Follow Tandem Diabetes
Care on LinkedIn at
https://www.linkedin.com/company/tandemdiabetes.
© 2024 Tandem Diabetes Care, Inc. All rights reserved. Tandem
Diabetes Care, the Tandem logo, Control-IQ, and t:slim X2 are
either registered trademarks or trademarks of Tandem Diabetes Care,
Inc. in the United States and/or other countries.
Important Safety Information – FreeStyle Libre
Systems
Failure to use FreeStyle Libre systems as instructed in labeling
may result in missing a severe low or high glucose event and/or
making a treatment decision, resulting in injury. If glucose
reading and alarms (if enabled) do not match symptoms or
expectations, use a fingerstick value from a blood glucose meter
for treatment decisions. Seek medical attention when appropriate or
contact Abbott at 855-632-8658 or
freestyle.abbott/us-en/safety-information.html for safety info.
The sensor house, FreeStyle, Libre, and related brand marks are
marks of Abbott and used with permission.
Responsible use of Control-IQ technology
Control-IQ technology does not prevent all highs and lows. Users
must still bolus for meals and actively manage their diabetes.
Visit tandemdiabetes.com/safetyinfo for additional important safety
information.
Important Safety Information – t:slim X2 Insulin Pump
RX ONLY. The t:slim X2 pump and Control-IQ technology are
intended for single patient use. The t:slim X2 pump and Control-IQ
technology are indicated for use with NovoLog or Humalog U-100
insulin. t:slim X2 insulin pump: The t:slim X2 insulin pump with
interoperable technology is intended for the subcutaneous delivery
of insulin, at set and variable rates, for the management of
diabetes mellitus in people requiring insulin. The pump is able to
reliably and securely communicate with compatible, digitally
connected devices, including automated insulin dosing software, to
receive, execute, and confirm commands from these devices. The pump
is indicated for use in individuals 6 years of age and greater.
Control-IQ technology: Control-IQ technology is intended for use
with compatible integrated continuous glucose monitors (iCGM, sold
separately) and alternate controller enabled (ACE) pumps to
automatically increase, decrease, and suspend delivery of basal
insulin based on iCGM readings and predicted glucose values. It can
also deliver correction boluses when the glucose value is predicted
to exceed a predefined threshold. Control-IQ technology is intended
for the management of Type 1 diabetes mellitus in persons 6 years
of age and greater.
WARNING: Control-IQ technology should not be used by
anyone under the age of 6 years old. It should also not be used in
patients who require less than 10 units of insulin per day or who
weigh less than 55 pounds.
Control-IQ technology is not indicated for use in pregnant
women, people on dialysis, or critically ill patients. Do not use
Control-IQ technology if using hydroxyurea. Users of the t:slim X2
pump and Control-IQ technology must: use the insulin pump, CGM, and
all other system components in accordance with their respective
instructions for use; test blood glucose levels as recommended by
their healthcare provider; demonstrate adequate carb-counting
skills; maintain sufficient diabetes self-care skills; see
healthcare provider(s) regularly; and have adequate vision and/or
hearing to recognize all functions of the pump, including alerts,
alarms, and reminders. The t:slim X2 pump must be removed before
MRI, CT, or diathermy treatment. Visit
tandemdiabetes.com/safetyinfo for additional important safety
information.
Forward-looking Statements – Tandem Diabetes Care
This press release contains “forward-looking statements” within
the meaning of Section 27A of the Securities Act of 1933, as
amended, and Section 21E of the Securities Exchange Act of 1934, as
amended. These forward-looking statements relate to, among other
things, our ability to provide the software update for current
t:slim X2 pump users and the t:slim X2 pumps pre-loaded with
Freestyle Libre 2 Plus sensor integration software. These
statements are subject to numerous risks and uncertainties,
including our ability to sustain commercial scale manufacturing of
the t:slim X2 pumps pre-loaded with Freestyle Libre 2 Plus sensor
integration software, our ability to operate and maintain a system
to facilitate online training for existing t:slim X2 pump customers
upgrading their existing devices, and the risk that we may
encounter other challenges that may delay the availability of
t:slim X2 pumps pre-loaded with Freestyle Libre 2 Plus sensor
integration software. These and other risks are identified and
described in greater detail under the “Risk Factors” heading of our
most recent Annual Report on Form 10-K, Quarterly Reports on Form
10-Q, and other documents filed with the Securities and Exchange
Commission. Readers are cautioned not to place undue reliance on
these forward-looking statements, which speak only as of the date
of this release. Actual results could differ materially from those
anticipated or projected in the forward-looking statements. Tandem
undertakes no obligation to update or review any forward-looking
statement in this press release because of new information, future
events, or other factors.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240108667517/en/
Abbott Media Contract: Ashley Brune
Ashley.Brune@abbott.com
Abbott Investor Contact: Mike Comilla
michael.comilla@abbott.com
Tandem Diabetes Care Media Contact: 858-366-6900
media@tandemdiabetes.com
Tandem Diabetes Care Investor Contact: 858-366-6900
IR@tandemdiabetes.com
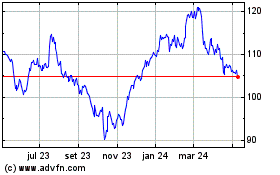
Abbott Laboratories (NYSE:ABT)
Gráfico Histórico do Ativo
De Jun 2024 até Jul 2024
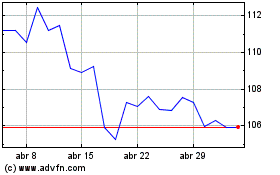
Abbott Laboratories (NYSE:ABT)
Gráfico Histórico do Ativo
De Jul 2023 até Jul 2024