Pfizer, EMD Serono: Bavencio Meets Primary Endpoint in Phase 3 Bladder Cancer Study
06 Janeiro 2020 - 9:52AM
Dow Jones News
By Colin Kellaher
Pfizer Inc. (PFE) and Merck KGaA's (MRK.XE) EMD Serono unit on
Monday said a phase III study of the immunotherapy Bavencio in
bladder cancer met its primary endpoint of overall survival at the
planned interim analysis.
The companies said patients with previously untreated locally
advanced or metastatic urothelial carcinoma whose disease didn't
progress on induction chemotherapy and who were randomized to
receive first-line maintenance therapy with Bavencio and best
supportive care lived significantly longer than those who received
best supportive care only.
The companies said Bavencio is the first immunotherapy to
significantly prolong overall survival in locally advanced or
metastatic urothelial carcinoma, the most common type of bladder
cancer, in a phase III trial.
Germany's Merck and New York-based Pfizer formed a strategic
alliance in 2014 to jointly develop and commercialize Bavencio.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
January 06, 2020 07:37 ET (12:37 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
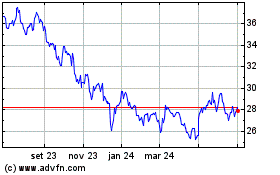
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Mar 2024 até Abr 2024
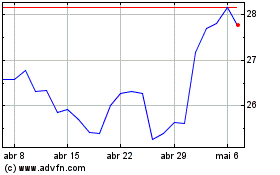
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Abr 2023 até Abr 2024