ARS Pharmaceuticals Shares Sink Premarket as FDA Rejects Neffy Spray
20 Setembro 2023 - 7:30AM
Dow Jones News
By Colin Kellaher
ARS Pharmaceuticals shares plunged in premarket trading
Wednesday after the U.S. Food and Drug Administration rejected its
proposed epinephrine nasal spray for the treatment of severe
allergic reaction despite an earlier endorsement from an FDA
advisory panel.
ARS, which plans to market the spray as "neffy," late Tuesday
said the FDA issued a so-called complete response letter,
indicating the agency won't approve the application in its current
form, and called on the San Diego company to complete a repeat-dose
study prior to approval.
ARS said it plans to appeal the decision, adding that it had
reached alignment with the FDA last month on post-market
requirements, including the repeat-dose study.
An FDA advisory panel in May recommended neffy approval based on
current data, without recommending additional studies. The FDA
usually follows the advice of its advisory committees, but it isn't
bound by the recommendations.
ARS said it expects to resubmit its application in the first
half of 2024, positioning it for potential approval in the second
half of the year.
The company said it expects to have about $195 million in cash,
equivalents and short-term investments at the time of the
anticipated launch of neffy if approved in the second half of
2024.
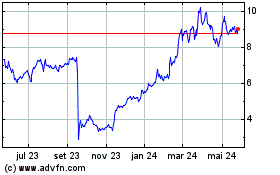
Shares of ARS, which closed Tuesday at $6.60, were recently down
46% to $3.55 in premarket trading.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
September 20, 2023 06:15 ET (10:15 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
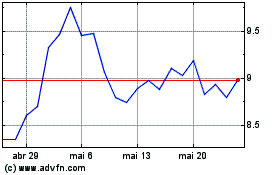
ARS Pharmaceuticals (NASDAQ:SPRY)
Gráfico Histórico do Ativo
De Mai 2024 até Jun 2024
ARS Pharmaceuticals (NASDAQ:SPRY)
Gráfico Histórico do Ativo
De Jun 2023 até Jun 2024