Merck, Pfizer, Astellas Get FDA OK For Expanded Use Of Keytruda Plus Padcev
15 Dezembro 2023 - 7:53PM
Dow Jones News
By Ben Glickman
Merck & Co., Pfizer and Astellas Pharma have received
approval from the U.S. Food and Drug Administration for using
Keytruda in combination with Padcev to treat a form of bladder
cancer.
Merck announced the FDA approval late Friday. The decision was
based on the results from a trial conducted in collaboration with
Seagen, now owned by Pfizer, and Astellas.
The new approval is specifically for Keytruda, Merck's
immunotherapy drug, and Padcev, Seagen and Astellas' antibody-drug
conjugate, in treating adult patients with locally advanced or
metastatic urothelial cancer.
Merck said the approval was nearly five months prior to the
FDA's target date.
Merck said the trial showed a statistically significant
improvement in its major efficacy endpoints of overall survival and
progression-free survival when compared to platinum-based
chemotherapy.
Write to Ben Glickman at ben.glickman@wsj.com
(END) Dow Jones Newswires
December 15, 2023 17:38 ET (22:38 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
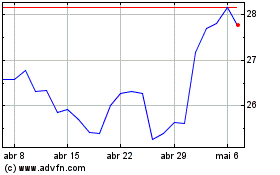
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
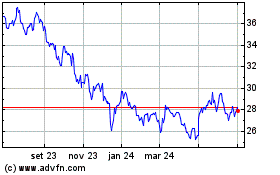
Pfizer (NYSE:PFE)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024