Akero Therapeutics Appoints Scott Gangloff as Chief Technology Officer
30 Abril 2024 - 9:00AM

Akero Therapeutics, Inc. (Nasdaq: AKRO), a clinical-stage company
developing transformational treatments for patients with serious
metabolic disease, today announced the appointment of Scott
Gangloff as Chief Technology Officer.
"I am delighted to welcome Scott to Akero. His depth of
experience leading drug manufacturing for both national and
multi-national biopharmaceutical organizations enriches our company
with an invaluable perspective during the late-stage clinical
development of efruxifermin," remarked Andrew Cheng, M.D., Ph.D.,
president and chief executive officer of Akero. "The entire
leadership team looks forward to partnering with Scott to advance
our commercialization and manufacturing strategy as we progress our
Phase 3 trials and work toward providing meaningful treatment
options to patients with MASH as quickly as possible."
Scott brings a strong track record of success in
biopharmaceutical development and manufacturing to Akero, with
nearly 30 years of hands-on experience in strategy, business and
technical development, and clinical and commercial supply chain
activities. Over the course of his career, he has led teams through
numerous BLAs, and successful product approvals in both the US and
Global markets. Most recently, Scott served as Vice President,
Global Biopharmaceutical Development, at Incyte Corporation. In
this role, he was responsible for setting the CMC vision and
strategy for all large molecules, technology transfer strategy
throughout product life cycles, post-approval transfer strategy and
implementation at the company’s internal commercial manufacturing
facility in Switzerland, as well as oversight of the end-to-end
supply chain from pre-clinical to commercial product launch. He
previously held various positions at Oncobiologics (now Outlook
Therapeutics), Bristol Myers Squibb, and Jacobs Engineering. Scott
earned his Master of Engineering degree in Chemical Engineering
from Lehigh University.
"I am eager to collaborate with this exceptional team to advance
the product development and manufacture of efruxifermin, currently
in Phase 3 trials for the treatment of MASH,” said Scott. “Backed
by encouraging scientific evidence, EFX is one of the most
promising investigational therapeutic solutions currently in
development for individuals with MASH. I look forward to partnering
with the team during this critical stage to prepare the
manufacturing process and associated supply chain of the
combination drug-device product for potential regulatory
authorization application and subsequent commercialization.”
About EfruxiferminEfruxifermin (EFX), Akero’s
lead product candidate for MASH, is a differentiated Fc-FGF21
fusion protein that has been engineered to mimic the balanced
biological activity profile of native FGF21, an endogenous hormone
that alleviates cellular stress and regulates metabolism throughout
the body. EFX appears to reduce liver fat and inflammation, reverse
fibrosis, increase insulin sensitivity and improve lipids. This
holistic approach offers the potential to address the complex,
multi-system disease state of MASH, including improvements in
lipoprotein risk factors linked to cardiovascular disease – the
leading cause of death in MASH patients. EFX is designed to offer
convenient once-weekly dosing and has been generally well tolerated
in clinical trials to date.
About MASHMASH is a serious form of MASLD that
is estimated to affect more than 17 million Americans. MASH is
characterized by an excessive accumulation of fat in the liver that
causes stress and injury to liver cells, leading to inflammation
and fibrosis, which can progress to cirrhosis, liver failure,
cancer and eventually death. MASH is the fastest growing cause of
liver transplants and liver cancer in the US and Europe.
About Akero TherapeuticsAkero Therapeutics
is a clinical-stage company developing transformational treatments
for patients with serious metabolic diseases marked by high unmet
medical need, including MASH. Akero's lead product candidate, EFX,
is currently being evaluated in two ongoing Phase 3 clinical
trials: the SYNCHRONY Histology study in patients with
pre-cirrhotic MASH (F2-F3 fibrosis) and the SYNCHRONY Real-World
study in patients with MASH or MASLD. A third clinical trial, the
SYNCHRONY Outcomes study in patients with cirrhosis due to MASH, is
expected to be initiated in the second quarter of 2024. The Phase 3
SYNCHRONY program builds on the results of two Phase 2b clinical
trials, the HARMONY study in patients with pre-cirrhotic MASH and
the SYMMETRY study in patients with cirrhosis due to MASH. Akero is
headquartered in South San Francisco. Visit us at akerotx.com and
follow us on LinkedIn and Twitter for more information.
Forward Looking Statements Statements contained
in this press release regarding matters that are not historical
facts are "forward-looking statements" within the meaning of the
Private Securities Litigation Reform Act of 1995. Because such
statements are subject to risks and uncertainties, actual results
may differ materially from those expressed or implied by such
forward-looking statements, including, but not limited to,
statements regarding Akero’s business plans and objectives,
including future plans or expectations for EFX, the therapeutic
effects of EFX; the timing and initiation of Akero’s Phase 3
SYNCHRONY program; and upcoming milestones; and the anticipated
contribution of Mr. Gangloff to its operations and progress. Any
forward-looking statements in this press release are based on
management's current expectations of future events and are subject
to a number of risks and uncertainties that could cause actual
results to differ materially and adversely from those set forth in
or implied by such forward-looking statements. Risks that
contribute to the uncertain nature of the forward-looking
statements include: the success, cost, and timing of Akero’s
product candidate development activities and planned clinical
trials; Akero’s ability to execute on its strategy; positive
results from a clinical study may not necessarily be predictive of
the results of future or ongoing clinical studies; regulatory
developments in the United States and foreign countries; Akero’s
ability to fund operations; as well as those risks and
uncertainties set forth more fully under the caption "Risk Factors"
in Akero’s most recent Annual Report on Form 10-K and Quarterly
Report on Form 10-Q, as filed with the Securities and Exchange
Commission (SEC) as well as discussions of potential risks,
uncertainties and other important factors in Akero’s other filings
and reports with the SEC. All forward-looking statements contained
in this press release speak only as of the date on which they were
made. Akero undertakes no obligation to update such statements to
reflect events that occur or circumstances that exist after the
date on which they were made.
Investor Contact:Christina
Tartaglia212.362.1200IR@akerotx.com
Media Contact:Sarah
O’Connell732.456.0092soconnell@vergescientific.com
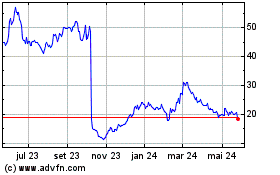
Akero Therapeutics (NASDAQ:AKRO)
Gráfico Histórico do Ativo
De Abr 2024 até Mai 2024
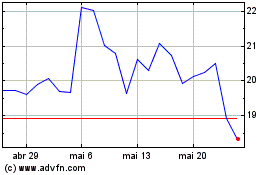
Akero Therapeutics (NASDAQ:AKRO)
Gráfico Histórico do Ativo
De Mai 2023 até Mai 2024